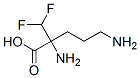
2-(Difluoromethyl)-DL-ornithine
- Product Name2-(Difluoromethyl)-DL-ornithine
- CAS70050-56-5
- MFC6H12F2N2O2
- MW182.17
- EINECS
- MOL File70050-56-5.mol
Usage And Synthesis
ChEBI: Eflornithine is a fluoroamino acid that is ornithine substituted by a difluoromethyl group at position 2. It has a role as a trypanocidal drug. It is a fluoroamino acid and an alpha-amino acid. It is functionally related to an ornithine.
Cultured bloodstream trypomastigotes of T. brucei are relatively
insensitive, but high doses are effective against bloodstream
and CNS infections of T. brucei brucei and T. brucei
gambiense in rodents, provided a strong antibody response
is also present. Eflornithine entry into the CNS can be
enhanced with suramin. T. brucei rhodesiense
infections do not respond. Synergy with some arsenicals has
been demonstrated.
Eflornithine is active against P. falciparum in experimental models and against Leishmania promastigotes and Giardia lamblia in culture.
Eflornithine is active against P. falciparum in experimental models and against Leishmania promastigotes and Giardia lamblia in culture.
Acquired resistance in T. brucei gambiense in West Africa has
not been reported. East African T. brucei rhodesiense strains
are innately less sensitive. Reported treatment failures are
thought to be associated with severity of disease.
An analog of ornithine, formulated as the hydrochloride for
intravenous infusion. It is freely soluble in water.
Oral absorption: 55%
Cmax 10 mg/kg oral: c. 7 mg/L after 4 h
200 mg/kg intravenously: 15.9 mg/L (87.5 nmol/mL) (mean)
Plasma half-life: 3.3 h
Volume of distribution: 0.34 L/kg
Plasma protein binding: Very low
Renal clearance is 83%, with most eliminated unchanged. In a study in Zaire the mean serum concentration in children under 12 years old was half that of adults, probably due to more rapid renal clearance. CNS penetration is good in adult patients, with a CSF:plasma ratio of 0.91 at the end of administration for 14 days. However, the CSF:plasma ratio in children under 12 years old was 0.58. Relapses have been recorded in patients in whom CSF levels dropped below 9 mg/L (50 nmol/mL) at the end of treatment.
Cmax 10 mg/kg oral: c. 7 mg/L after 4 h
200 mg/kg intravenously: 15.9 mg/L (87.5 nmol/mL) (mean)
Plasma half-life: 3.3 h
Volume of distribution: 0.34 L/kg
Plasma protein binding: Very low
Renal clearance is 83%, with most eliminated unchanged. In a study in Zaire the mean serum concentration in children under 12 years old was half that of adults, probably due to more rapid renal clearance. CNS penetration is good in adult patients, with a CSF:plasma ratio of 0.91 at the end of administration for 14 days. However, the CSF:plasma ratio in children under 12 years old was 0.58. Relapses have been recorded in patients in whom CSF levels dropped below 9 mg/L (50 nmol/mL) at the end of treatment.
2-(Difluoromethyl)-DL-ornithine has been used speculatively for treatment of Pn. jirovecii infections in AIDS patients. Co-administration with oral nifurtimox has been added to the World Health Organization (WHO) List of Essential Medicines for second-stage sleeping sickness caused by T. brucei gambiense.
Osmotic diarrhea and bone marrow suppression are common,
and up to 50% of sleeping sickness patients develop
leukopenia. Reversible anemia and thrombocytopenia have
been observed. Convulsions and seizures, different from those
observed in melarsoprol-induced encephalopathy, have been
reported in 4–18% of treated sleeping sickness patients but
not in patients treated for Pn. jirovecii pneumonia. This difference
might be due to the CNS inflammation associated with
sleeping sickness.
2-(Difluoromethyl)-DL-ornithine Supplier
Tel +86-400-002-6226 +86-13028896684;
Email sales@rrkchem.com
PROMPT×
PROMPT
The What'sApp is temporarily not supported in mainland China
The What'sApp is temporarily not supported in mainland China
Cancel
Determine