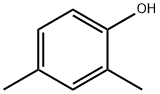
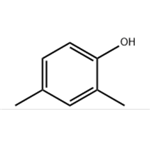
2,4-dimethylphenol (also named as 2,4-xylenol) is a substituted phenol which occurs naturally. It can be also derived from the cresol fraction of petroleum or coal tars by fractional distillation and extraction with aqueous alkaline solution. It can be used as a feedstock for the production of phenolic antioxidants, disinfectants, solvents, pharmaceuticals, insecticides, fungicides, plasticizers, rubber chemicals, polyphenylene oxide, dyestuffs, wetting agents, and as an additive or constituent of gasolines, lubricants, and cresylic acid. It is an allowed liquid smoke flavoring agent in the EU.
- J. P. Ghosh, K. E. Taylor, J. K. Bewtra, N. Biswas, Laccase-catalyzed removal of 2,4-dimethylphenol from synthetic wastewater: Effect of polyethylene glycol and dissolved oxygen, Chemosphere, 2008, vol. 71, pp. 1709-1717
- G. W. Holcombe, G. L. Phipps, J. T. Fiandt, Effects of phenol, 2,4-dimethylphenol, 2,4-dichlorophenol, and pentachlorophenol on embryo, larval, and early-juvenile fathead minnows (Pimephales promelas), Archives of Environmental Contamination and Toxicology, 1982, vol. 11, pp. 73-78
- A. Giri, Z. Zelinkova, T. Wenzl, Experimental design-based isotope-dilution SPMEGC/MS method development for the analysis of smoke flavouring products, Food Additives & Contaminants: Part A, 2007, vol. 34, pp. 2069-2084
2,4-DMP is a combustible, colorless, crystalline solid. The 2,4-isomer is 1 of 5 isomers of this formula.Molecular weight= 122.18; Boiling point 212℃; Freezing/Melting point = 2728℃; Flash point = 110℃. HazardIdentification (based on NFPA-704 M Rating System):Health 1, Flammability 2, Reactivity 0. Soluble in water.
2,4-Dimethylphenol is a clear colorless to yellow liquid or needle crystals. miscible with alcohol and ether, slightly soluble in water.It has a moderate mammalian oral toxicity and is a recognised irritant.
Colorless solid, slowly turning brown on exposure to air.
2,4-dimethylphenol is a fungicide and disinfectant with a variety of agricultural uses. It is also used in making wetting agent; dyestuffs; phenolic antioxidants; pharmaceuticals; rubber chemicals; lubricant ; gasoline additive and plasticizers.
It is used as a perfuming agent in cosmetic industry. Also used in the production of high-viscosity phosphate esters, as a feedstock for hindered phenol antioxidant and specialty modified phenolic resin manufacture.
synthesis of 2,4-dimethylphenol: 2,4-Dimethylphenol is obtained by sulfonation, salting out, alkali melting and acidification of m-xylene.
ChEBI: 2,4-xylenol is a member of the class of phenols that phenol substituted by methyl groups at positions 2 and 4. It has a role as a disinfectant and a volatile oil component. It is a member of phenols and an aromatic fungicide. It derives from a hydride of a m-xylene.
2,4-dimethylphenol appears as colorless crystals or clear, dark amber liquid.
2,4-Dimethylphenol is a very weak acid (pKa = 10.6) . Incompatible with acid chlorides, acid anhydrides, bases and oxidizing agents. Corrodes steel, brass, copper and copper alloys .
2,4-Dimethylphenol is expected
to be an irritant of the eyes, mucous membranes,
and skin, by analogy to other phenols.
The oral LD50 for rats was 3.2 g/kg; the
dermal LD50 in mice was 1.04 g/kg.
2,4-Dimethylphenol is probably combustible.
Poison by intravenous
and intraperitoneal routes. Moderately toxic
by ingestion and skin contact. Questionable
carcinogen with experimental carcinogenic
data. When heated to decomposition it
emits acrid smoke and irritating fumes. See
also other xylenol entries.
AgriculturalChemical; Tumorigen, Drug. 2,4-DMP finds use commercially as an important chemical feedstock or constituent forthe manufacture of a wide range of commercial productsfor industry and agriculture. 2,4-Dimethylphenol is used inthe manufacture of phenolic antioxidants, disinfectants, solvents, pharmaceuticals, insecticides, fungicides, plasticizers,rubber chemicals, polyphenylene oxide wetting agents, anddyestuffs; and is an additive or constituent of lubricants,gasolines, and cresylic acid. 2,4-Dimethylphenol (2,4-DMP)is a naturally occurring substituted phenol derived from thecresol fraction of petroleum or coal tars by fractional distillation and extraction with aqueous alkaline solutions. It isthe cresylic acid or tar acid fraction of coal tar. Workersinvolved in the fractionation and distillation of petroleum orcoal, and coal tar products comprise one group at risk.Workers who are intermittently exposed to certain commercial degreasing agents containing cresol may also be at risk.Cigarette and marijuana smoking groups and those exposedto cigarette smoke inhale microgram quantities of 2,4-demthylphenol.
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. If convulsions are not present, give a glass ortwo of water or milk to dilute the substance. Assure that theperson’s airway is unobstructed and contact a hospital orpoison center immediately for advice on whether or not toinduce vomiting
2,4-Dimethylphenol was tested for mutagenicity
in the Salmonella microsome preincubation
assay using the standard protocol of the
National Toxicology Program and five strains
of Salmonella; results were negative.
The ACGIH has not established a threshold
limit value (TLV) for 2,4-dimethylphenol.
Thomas and Delfino (1991) equilibrated contaminant-free groundwater collected from Gainesville, FL with individual fractions of three individual petroleum products at 24–25 °C for
24 h. The aqueous phase was analyzed for organic compounds via U.S. EPA approved test method
625. Average 2,4-dimethylphenol concentrations reported in water-soluble fractions of unleaded
gasoline, kerosene, and diesel fuel were 50, 99, and 108 μg/L, respectively. 2,4-Dichlorophenol
may also enter groundwater by leaching from coal tar, asphalt runoff, plastics, and pesticides
(quoted, Verschueren, 1983).
Biological. When 2,4-dimethylphenol was statically incubated in the dark at 25 °C with yeast
extract and settled domestic wastewater inoculum, significant biodegradation with rapid
adaptation was observed. At concentrations of 5 and 10 mg/L, 100 and 99% biodegradation,
respectively, were observed after 7 d (Tabak et al., 1981).
Photolytic. 2,4-Dimethylphenol absorbs UV light at a maximum wavelength of 277 nm
(Dohnal and Fenclová, 1995).
Chemical/Physical. Wet oxidation of 2,4-dimethylphenol at 320 °C yielded formic and acetic
acids (Randall and Knopp, 1980). 2,4-Dimethylphenol will not hydrolyze because there is no
hydrolyzable functional group (Kollig, 1993).
Color Code—Blue: Health Hazard/Poison: Storein a secure poison location. Prior to working with thischemical you should be trained on its proper handling andstorage. Store in tightly closed containers in a cool, wellventilated area away from oxidizers and sources of ignition
This compound requires a shipping label of“POISONOUS/TOXIC MATERIALS” for xylenols. It fallsin Hazard Class 6.1 and Packing Group II.
The initial threshold screening level (ITSL) for 2,4-dimethylphenol is 70 μg/m3 based on an annual averaging time.