OFF-WHITE TO SLIGHTLY GREY GRANULAR CRYST. POWDER
A sulfanilamide derivative of Chitosan
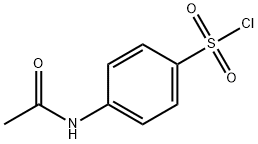
Intermediate in the preparation of sulfanilamide and its derivatives.
N-Acetylsulfanilyl chloride is usedin the preparation of sulfanilamide and its derivatives which are intermediates to produce sulfa drugs. They are used in the prevention and treatment of bacterial infections, diabetes mellitus, edema, hypertension, and gout. It is also used as a Pharma raw material. N-Acetylsulfanilyl chloride widely used in the fields of dye, medicine, mainly used for preparation of Sulfanilamide, Sulfanilylureal, Sulfatolamide, Sulphathiourea , Sulfaguanidine and Sulfacetamide and etc.
Acetanilide is introduced into stirred chlorosulfuric acid solution at 20 ℃. The solution is then heated to 55 ℃ and kept at this temperature for 1.5 h. The solution is cooled to 20 ℃ and introduced as a thin jet into water, the temperature of which is kept at 0 – 5 ℃ by external cooling. The precipitated sulfochloride is filtered and washed with cold water until neutral to Congo Red. Yield is 80 – 82%. The crude product contains about 60% water; the dried product melts at 144 – 147 ℃. About 1 – 2% of bis(acetylaminophenyl) sulfone is obtained as a byproduct. If thionyl chloride is added, less chlorosulfuric acid is needed because it can be replaced partly byoleum. The product is stable for several days if kept in a cool place. It is generally used directly in the moist state. 4-(Acetylamino)benzenesulfonyl chloride is an important intermediate in the manufacture of sulfonamides and in the synthesis of parabase ester.
Biotechnological Production
After more than three decades of strain and process optimization, the 2KGA fermentation
by K. vulgare has reached a performance level that makes it increasingly
difficult to achieve further cost-relevant improvements. Instead, opportunities can
be seen in the succeeding step of 2KGA rearrangement to ascorbic acid, which still
follows the same concept as laid out in the 1930s by Reichstein and Grüssner. This
chemical step contributes significantly to the overall process costs. A process
Industrial Production of L-Ascorbic Acid (Vitamin C) and D-Isoascorbic Acid 171
concept that could convert sorbitol directly to ascorbic acid would therefore be
most attractive. In theory, this could build on the established 2KGA fermentation
with an enzyme-catalyzed 2KGA to Asc rearrangement (2,6-hemiacetal to 1,4-
lactone) as extension. Ab initio energy calculations as well as experimental results
(own unpublished results) indicate that in aqueous environment, Asc is thermodynamically
far more stable than 2KGA and (nearly) quantitative conversion
should be possible. However, no enzyme efficiently catalyzing this reaction has so
far been identified. The few publications of enzyme catalysis for this reaction so far
shows only trace activity and no significant improvements have been
reported. 2KGA may represent a kinetic trap in an aqueous environment and biotechnological
reaction pathways all the way to Asc may need to avoid 2KGA.
Accordingly, 2KGA is also not part of natural biosynthetic routes, where Asc
formation directly results from the oxidation of precursor molecules
with appropriately preformed 1,4-lactone linkage (L-gulono-1,4-lactone in animals,
L-galactono-1,4-lactone in plants). Enzymes converting L-gulono-1,4-lactone to
Asc are also known from bacteria, even from Ketogulonicigenium. The biochemical
description of the Ketogulonicigenium enzyme indicates that it belongs to
the family of heterotrimeric periplasmic flavohemoproteins, of which several can
be found in the published Ketogulonicigenium genomes. Besides sharing the same
FAD cofactor, these enzymes bear no similarity to the mammalian gulono-1,4-
lactone dehydrogenase. The use of these natural or nature-like Asc-forming
enzymatic steps in biotechnological production processes is so far precluded by the
rare nature of these L-sugar-derived lactone precursor molecules and the lack of
efficient production methods for these compounds. It was, therefore, a tantalizing
discovery when Asc formation directly from L-sorbosone, the intermediate of the
efficient 2KGA formation route, was identified in those two species already in the
focus for 2KGA production for decades: K. vulgare and G. oxydans.
Besides an earlier report of L-sorbosone to Asc activity derived from plant tissue
, which did not see consolidating follow-ups, the above observations are the
first evidence of biological Asc formation from a molecule other than a 1,4-lactone.
L-hydroxyproline has been derivatized with N-acetylsulfanilyl chloride and 5-chlorovaleric acid during the synthesis of the haptens HP1 and HP2.
Flammability and Explosibility
Not classified
A poison by intraperitoneal route.Moderately toxic by ingestion. When heated todecomposition it emits toxic vapors of NOx, SOx, and Cl.
Crystallise the chloride from toluene, CHCl3, or ethylene dichloride. [Beilstein 14 IV 2703.]