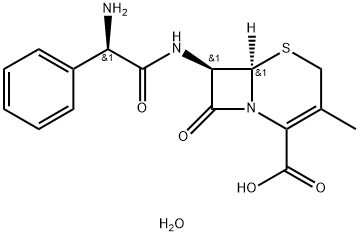
Cephalexin was first synthesized in 1967 by Glaxo Research Laboratories and first produced on an industrial scale by Eli Lilly & Co. in the same year. It is a deacetoxylated derivative of cephaloglycin that is not metabolized in vivo. When administered orally, it shows a much higher serum concentration and much lower tendency to induce diarrhea than cephaloglycin. Cephalexin has been used widely and is the most popular orally active antibiotic in the world for treatment of respiratory tract, urinary tract, surgical, ear and nose, and other infections caused by Staphylococcus, Streptococcus, Escherichia coli, Klebsiella, Enterobacter, and Proteus.
Semi-synthetic cephalosporin antibiotic.
Cephalosporin antibacterial.
A semisynthetic cephalorsporin antibiotic.
ChEBI: The hydrate of cephalexin.
To a 1 liter flask containing dimethylformamide at 0°C, was added 24.8 g
sodium N-(2-methoxycarbonyl-1-methylvinyl)-D-α-phenylglycine (prepared
from sodium D-α-phenylglycine and methyl acetoacetate). The mixture was
cooled to -40°C and methyl chloroformate (7.5 ml) and dimethylbenzylamine
(0.26 ml) added. After stirring for 25 minutes, p-nitrobenzyl 7-
aminodesacetoxycephalosporanate (32.8 g) in the form of its hydrochloride
salt was added, followed by triethylamine (12.1 ml) and dimethylformamide
(140 ml) over a period of 20 minutes. The reaction mixture was stirred for 2
hours at -25°C to -35°C, then warmed to 0°C and water (32 ml) added. To
the resultant solution, hydrochloric acid (54 ml) was added followed by zinc
(21.8 g) in portions over a period of 5 minutes, the temperature being
maintained at 5°C to 10°C. Further hydrochloric acid (35 ml) was added and
the solution stirred at 15°C to 20°C for 7 hours.
The pH was adjusted to 3.3 with triethylamine and
semicarbazidehydrochloride (9.5 g) added. The mixture was brought back to
pH 3 with further triethylamine, then stirred for 30 minutes at pH 3. The
resultant mixture was adjusted slowly over 4 hours to pH 6.8 by addition of
triethylamine, seeding being carried out when pH 4.5 was reached. The precipitated cephalexin was filtered off, washed with dimethylformamide (200
ml) and the cephalexin recovered, yield 75%.
Keflex (Panixine (Ranbaxy).