Pyrophosphoric acid may be prepared by heating orthophosphoric acid at 215°C:
2H3PO4 → H4P2O7 + H2O
The acid solution in pure form can be obtained by ion exchange, passing an aqueous solution of sodium pyrophosphate, Na4P2O7, through a suitable cation exchange column.
The acid has four replacable H+ ions. Its dissociation constants indicate that two H+ ions are strongly acidic while the other two protons are weakly acidic. The first dissociation constant especially is very large:
H4P2O7 + H2O ↔ H3O+ + H3P2O7¯ Ka1 ~ 10–1
H3P2O7¯+ H2O ↔ H3O+ + H2P2O72– Ka2 ~ 1.5x10–2
PHOSPHORIC ACID, PYRO 701H2P2O72¯ + H2O ↔ H3O+ + HP2O73– Ka3 ~ 2.7x10–7
HP2O73¯ + H2O ↔ H3O+ + P2O74¯ Ka4 ~ 2.4x10–10
Pyrophosphoric acid forms acid salts, such as NaH3P2O7 and Na2H2P2O7.
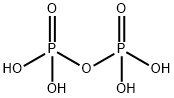
Pyrophosphoric acid, also known under the name
diphosphoric acid, is colorless, odorless, hygroscopic
and is soluble in water, diethyl ether and ethyl alcohol.
It is produced from phosphoric acid by dehydration.
Pyrophosphoric acid slowly hydrolyzes in the presence
of water into phosphoric acid.
H4P2O7 +H2O2H3PO4
Upon heating, phosphoric acid loses water, being
converted successively, and to some extent concurrently,
into the pyro- and meta-acids:
2H3PO4H4P2O7 +H2O
nH3PO4(HPO3)n + [H2O]n
White to very pale yellow glassy solid
Colorless needles or liquid; hygroscopic. Crystallizes in two anhydrous forms: a metastable form melting at 54.3°C and a second and more stable form melting at 71.5°C; extremely soluble in cold water, reacting very slowly to form phosphoric acid; decomposing much faster in hot water; very soluble in alcohol and ether.
Catalyst, manufacture of organic phosphate
esters, metal treatment, stabilizer for organic per-
oxides.
Pyrophosphoric Acid has been shown to enhance the rate of pyrophosphate hydrolysis by nonenzymatic catlysis and by inorganic pyrophoshatase. Pyrophosphoric Acid can be used for the preparation of vanadyl pyrophosphato methoxide cluster anion. Pyrophosphoric Acid is used in anion binding of inorganic phosphates due to is ability to form complexes.
According to early investigators, the pyro-acid is
formed as low as 100 °C, and the conversion is complete
between 255 and 260 °C. Phosphorous oxychloride
reacts with phosphoric acid in the following manner:
5H3PO4 + POC133H4P2O7 + 3HC1
A mixture of the ortho- and meta-acids may be
condensed together to form the pyro-acid by heating on the water bath:
nH3PO4 +[HPO3]n[H4P2O7]n
ChEBI: An acyclic phosphorus acid anhydride obtained by condensation of two molecules of phosphoric acid.