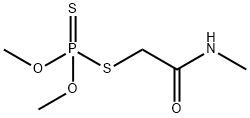
Dimethoate is a grey-white crystalline solid at room temperature. It is sparingly soluble in water, soluble in methanol and cyclohexane, but very soluble in chloroform and benzene. It has been classified by the U.S. EPA under GUP. Dimethoate is used extensively for the control of crop pests such as mites, aphids, thrips, plant hoppers, white-flies, and a wide range of other insects that damage, crops, fruits, vegetables, and ornamental plants. Dimethoate is also used for the control of cattle grubs that infect livestock. Thermal decomposition of dimethoate is highly hazardous due to the release of fumes of dimethylsulphide, methyl mercaptan, carbon monoxide, carbon dioxide, phosphorus pentoxide, and nitrogen oxides.
[1]Engenheiro, E. L., et al. "Influence of dimethoate on acetylcholinesterase activity and locomotor function in terrestrial isopods." Environmental Toxicology & Chemistry 24.3(2005):603.
[2]John, Susan, et al. "Protective effect of vitamin E in dimethoate and malathion induced oxidative stress in rat erythrocytes." The Journal of nutritional biochemistry 12.9 (2001): 500-504.
[3]Sharma, Yukti, et al. "Dimethoate-induced effects on antioxidant status of liver and brain of rats following subchronic exposure." Toxicology 215.3 (2005): 173-181.
Dimethoate
is colorless crystalline substance, mp 49 C, bp
117 C/0.1 mm Hg, vp 0.25 mPa (25 C). The solubility in
water is 23.8 g/L at 20 C and pH 7. It is readily soluble
in polar organic solvents. Log Kow = 0.7. Dimethoate
is relatively stable in aqueous media at pH 2–7 and
hydrolyzed in alkaline media; DT50 (20 C) at pH 9 is 12 d.
Dimethoate is a gray-white crystalline solid at room temperature. It is sparingly soluble in
water, soluble in methanol and cyclohexane, but very soluble in chloroform, benzene. It has
been classifi ed by the US EPA under GUP. Dimethoate is used extensively for the control
of crop pests, such as mites, aphids, thrips, plant-hoppers, white-fl ies, and a wide range
of other insects that damage, crops, fruits, vegetables, and ornamental plants. Dimethoate
is also used for the control of cattle grubs that infect livestock. Thermal decomposition of
dimethoate is highly hazardous owing to the release of fumes of dimethylsulfi de, methyl
mercaptane, carbon monoxide, carbon dioxide, phosphorus pentoxide, and nitrogenoxides.
Dimethoate is used to control of a wide range of insect and mite
pests in many crops. It is also used to control flies in animal houses.
An organophosphate insecticide. It is an anticholinesterase which disables cholinesterase, an enzyme essential for central nervous system function. Neurotoxic in humans.
insecticide, cholinesterase inhibitor
Systemic and contact organophosphorus insecticide and acaricide used to control thrips and red spider mites on many agricultural crops, sawies on apples and plums, wheat bulb and olive ies.
Systemic and contact insecticide.
ChEBI: A monocarboxylic acid amide that is N-methylacetamide in which one of the hydrogens of the methyl group attached to the carbonyl moiety is replaced by a (dimethoxyphosphorothioyl)sulfanediyl group.
A white crystalline solid, with a camphor-like odor, white to grayish crystals for technical product. Dimethoate is a contact and systemic organophosphate insecticide effective against a broad range of insects and mites when applied on a wide range of crops. Dimethoate has not been produced in the U.S. since 1982.
Dimethoate is stable in aqueous solution but is hydrolyzed by aqueous alkali.
Dimethoate is incompatible with alkaline preparations. Dimethoate is slightly corrosive to iron. Dimethoate is incompatible with sulfur based formulations. . Organophosphates are susceptible to formation of highly toxic and flammable phosphine gas in the presence of strong reducing agents such as hydrides. Partial oxidation by oxidizing agents may result in the release of toxic phosphorus oxides.
Very toxic; the probable oral lethal dose in humans is between 50-500 mg/kg, or between 1 teaspoon and 1 ounce for a 70 kg (150 lb.) person. Dimethoate is a cholinesterase inhibitor, meaning it affects the central nervous system. Death is due to respiratory arrest arising from failure of respiratory center, paralysis of respiratory muscles, intense bronchoconstriction or all three.
Cholinesterase inhibitor; very toxic, exhibit-ing acute, delayed, and chronic toxicity;routes of entry—ingestion, skin contact,and inhalation; toxic symptoms include nausea, vomiting, diarrhea, excessive salivation,bronchoconstriction, and respiratory arrest;oral intake of 5–20 g may cause death toadult humans.
LD50 oral (mammal): 15 mg/kg
The oral LD50 in rat, however, is higher(within the range 150 mg/kg) than someother common organophosphorus insecti-cides, such as Nemaphos, Mevinphos, orCoumaphos.
Dimethoate is toxic to animals and humans. Occupational exposures cause poisoning with
symptoms that include, but are not limited to, sweating, headache, weakness, giddiness,
nausea, vomiting, stomach pains, blurred vision, pupillary constriction, slurred speech, and
muscle twitching. Workers repeatedly exposed to dimethoate have shown symptoms of
numbness, tingling sensations, incoordination, headache, dizziness, tremor, nausea, abdominal
cramps, diffi culty breathing or respiratory depression, slow heart beat, and speech
diffi culties. Prolonged exposures cause severe poisoning with adverse effects on the CNS,
leading to incoordination, slurred speech, loss of refl exes, weakness, fatigue, involuntary
muscle contractions, twitching, tremors of the tongue or eyelids, and eventually paralysis
of the body extremities and the respiratory muscles, psychosis, irregular heart beats, unconsciousness,
convulsions, coma, and death caused by respiratory failure or cardiac arrest.
As with other organophosphorus pesticides, container may explode in heat of fire. The temperature of storage should not exceed 70-80F. Keep away from sources of heat, flames, or spark-generating equipment. Unstable in alkaline solution. Hydrolyzed by aqueous alkali. Stable in aqueous solutions. The compound is stable for 2 years under environmental conditions if stored in undamaged, original containers.
Insecticide, Miticide: Dimethoate is used to kill mites and insects systemically and on contact. It is used against a wide range of
insects, including aphids, thrips, planthoppers, and whiteflies on ornamental plants, alfalfa, apples, corn, cotton,
grapefruit, grapes, lemons, melons, oranges, pears, pecans, safflower, sorghum, soybeans, tangerines, tobacco,
tomatoes, watermelons, wheat, and other vegetables. It is
also used as a residual wall spray in farm buildings for
house flies. Dimethoate has been administered to livestock
for control of botflies. Dimethoate is available in aerosol
spray, dust, emulsifiable concentrate, and ULV concentrate
formulations.
REBELATE®; CEKUTHOATE®;
CHIMIGOR 40®; CYGON 400®[C]; DEFEND®;
DAPHENE®; DANADIM®; DANADIM® PROGRESS;
DE-FEND®; DEMOS NF®; DEVIGON®; DICAP®;
DIMATE 267®; DIMET®; DIMETHOATE 40;
DIMETHOPGAN®; FERKETHION®; FOSTION
MM®; KENLOGO®; NUGOR®; PERFEKTION®;
ROGODAN®; ROGODIAL®; ROGOR®; ROXION®;
SEVIGOR®
This organophosphorus compound is used as a contact
and systemic insecticide and acaricide. It induced an
erythema-multiform-like contact dermatitis in a ware-
houseman in an agricultural consortium.
A deadly human
poison. Poison by ingestion, skin contact,
intraperitoneal, and subcutaneous routes.
Moderately toxic by intravenous route.
Human systemic effects: coma, dyspnea,fasciculations. Questionable carcinogen with
experimental carcinogenic data.
Experimental teratogenic and reproductive
effects. Human mutation data reported.
When heated to decomposition it emits very
toxic fumes of NOx、, POx, and SOx. See also
ESTERS.
Compound Description:Agricultural Chemical; Tumorigen, Mutagen;Reproductive Effector; Human Data. Dimethoate is acontact and systemic organophosphate insecticide effective against a broad range of insects and mites whenapplied on a wide range of crops. It has not been produced in the United States since 1982.
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Speed in removing material from skinis of extreme importance. Shampoo hair promptly if contaminated. Seek medical attention immediately. If thischemical has been inhaled, remove from exposure, beginrescue breathing (using universal precautions, includingresuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. Give large quantities of water and inducevomiting. Do not make an unconscious person vomit.Effects may be delayed; keep victim under medical observation. Obtain authorization and/or further instructions fromthe local hospital for administration of an antidote or performance of other invasive procedures.Note to physician: 1,10-trimethylenebis(4-formylpyridiniumbromide)dioxime (a.k.a TMB-4 dibromide and TMV-4) hasbeen used as an antidote for organophosphate poisoning.
Rats given diets with 0, 5, 25,
or 100 ppm dimethoate (0, 0.25, 1.25, or 5 mg/kg/day) for
2 years showed dose-related trends for spleen hemangiosarcoma;
combined spleen hemangioma and hemangiosarcoma;
and combined spleen hemangioma, hemangiosarcoma, and
skin hemangiosarcoma . There were also significant
differences in pair-wise comparisons between controls and
low-dose (0.25 mg/kg) or high-dose (5 mg/kg) groups for
spleen (hemangioma/hemangiosarcoma) and combined
tumors of spleen and skin hemangiosarcoma and lymph
angioma/angiosarcoma; and between low and high doses
for all tumors combined.
Soil. Duff and Menzer (1973) reported that in moist soils, dimethoate is converted to the oxygen analog, dimethoate carboxylic acid (dimethoxon) and two unidentified metabolites. The degradation rate of dimethoate in three different soils increased almost twofold with a 10°C increase in temperature (Kolbe et al., 1991). The reported half-lives of dimethoate in a humus-rich sandy soil, clay loam and heavy clay soil at 10 and 20°C are 15.3, 10.3, 15.5 days and 9.7, 4.8 and 8.5 days, respectively. Degradates included dimethoxon (O,O-dimethyl-S-(N-methylcarbamoylmethyl)phosphorothiolate) and unidentified polar compounds (Kolbe et al., 1991).
In a silt loam and sandy loam, reported Rf values were 0.42 and 0.45, respectively (Sharma et al., 1986).
Plant. In plants, oxidation/hydrolysis leads to the formation of the phosphorothioate. Other hydrolysis products in plants include O,O-dimethylphosphorodithioate and O,Odimethylphosphorophosphate which occurs via demethylation and hydrolytic clea
The half-life in Bermuda grass was reported to be 3.1 days (Beck et al., 1966). The disappearance half-lives of dimethoate on bean, tomato, cucumber and cotton plants were 4.3, 6.0, 3.8 and 3.3 days, respectively (Belal and Gomaa, 1979).
Surface Water. Though no products were identified, the half-life in raw river water was 8 weeks (Eichelberger and Lichtenberg, 1971). Photolytic. Dichlorvos should not undergo direct photolysis since it does not absorb UV light at wavele
Dimethoate is a systemic insecticide which is rapidly translocated to all
parts of the plant. Metabolism to omethoate (dimethoate oxon), the active
cholinesterase inhibitor, is a major pathway in both animals and plants.
Degradative metabolism is either via dealkylation to yield both des-O-
methyldimethoate and des-O-methylomethoate, followed by hydrolysis
or dealkylation to give mono- and di-methyl phosphate, phosphorothioate
and phosphorodithioate esters which are ultimately degraded to phosphate.
An additional mechanism is through hydrolysis of the amide
group to give dimethoate and omethoate carboxylic acids. An important
additional metabolic pathway is oxidative N-demethylation of dimethoate
and omethoate to give des-N-methyldimethoate and des-N-methy
lomethoate via their respective N-hydroxymethyl derivatives. It is possible that the N-demethylation route could precede amidase action
leading to dimethoate and omethoate carboxylic acids which are excreted
in mammals. The 'thioglycolate' part of the molecule is metabolised to
α-hydroxy-N-methylacetamide in mammals.
Color Code—Blue: Health Hazard/Poison: Storein a secure poison location. Store in tightly closed containers in a cool, well-ventilated area away from heat andstrong bases. Do not store solid Dimethoate above25-30℃/77-86°F. However, liquid solutions of dimethoate must be stored above 7℃/45°F. Keep away fromsources of heat, flames, or spark-generating equipment.Unstable in alkaline solution. Hydrolyzed by aqueous alkali.Stable in aqueous solutions. The compound is stable for 2years under environmental conditions if stored in undamaged, original containers.
Organophosphorus pesticides, liquid, toxic, n.o.s.require a “POISONOUS/TOXIC MATERIALS” label.They fall in Hazard Class 6.1 and Packing Group II.
The acute oral
LD50 for rats is 387 mg/kg. Inhalation LC50 (4 h) for rats
is >1.6 mg/L air. NOEL (2 y) for rats is 5 mg/kg diet
(0.25 mg/kg/d). ADI is 2 μg/kg (sum of dimethoate and its
oxon, i.e., omethoate). Oxidative desulfuration to form
omethoate, the active AChE inhibitor, occurs both in
mammals and plants. The main degradation routes are
O-demethylation and amide hydrolysis that is important
particularly for the selective species toxicity in animals.
The cleavage of the PS and SC linkages also occurs to a
considerable degree. Aerobic DT50 in soil is 2–4 d,whereas
it was 22 d under anaerobic conditions.
Dimethoate is stable in aqueous media in the pH range 2-7 but hydrolysed
under alkaline conditions (PM). Under acidic conditions the sole
product of hydrolysis is desmethyldimethoate (2), whereas in alkali both
desmethyldimethoate (2) and O,O-dimethylphosphorothioate (3) were
formed. These products are in accord with other studies on the effects of
pH on the hydrolysis of phospho-triesters, with dealkylation predominating
under acidic conditions and SN2 attack on phosphorus by OH- at
high pH (PSD, 1993).
Dimethoate is stable to photolysis. In an experiment to measure
the degradation of an aqueous solution of dimethoate, a sample of
[O-14C-methyl]dimethoate was irradiated with a xenon arc lamp for 15
days. The compound was extracted unchanged (93.4%) and no specific
degradation products were identified (PSD, 1993).
Strong bases (alkalis). Do not store solidabove 77-86°F/25-30℃. Liquid solutions must be storedabove 45°F/7℃. Strong oxidizers may cause release oftoxic phosphorus oxides. Organophosphates, in the presenceof strong reducing agents such as hydrides, may formhighly toxic and flammable phosphine gas. Keep awayfrom alkaline materials.