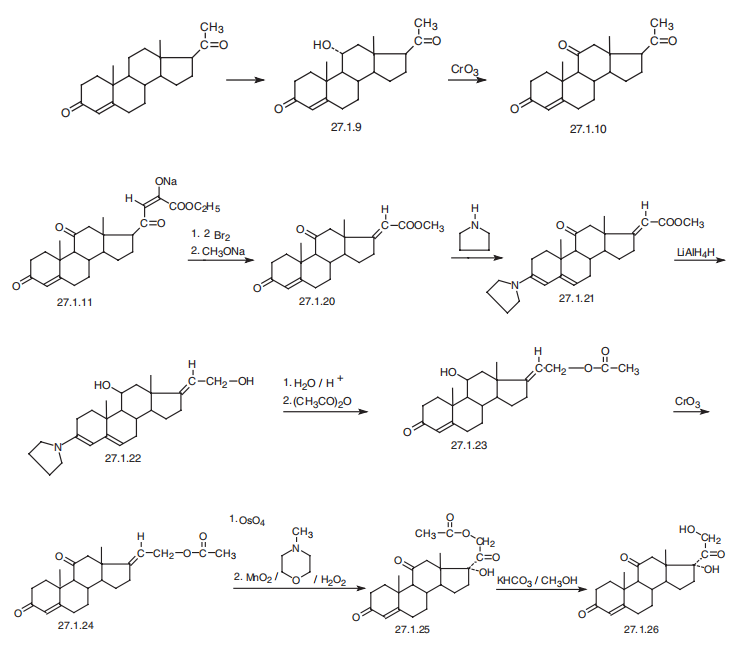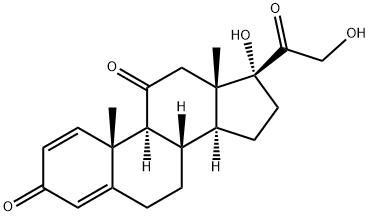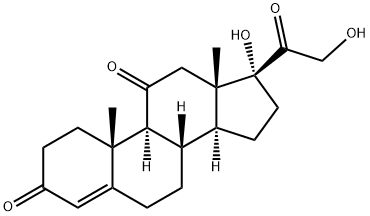
CORTISONE
- Product NameCORTISONE
- CAS53-06-5
- CBNumberCB6413305
- MFC21H28O5
- MW360.44
- EINECS200-162-4
- MDL NumberMFCD00003610
- MOL File53-06-5.mol
- MSDS FileSDS
Chemical Properties
| Melting point | 223-228 °C (dec.)(lit.) |
| alpha | D25 +209° (c = 1.2 in 95% alcohol); 25546 +269° (c = 0.125 in benzene); 25546 +248° (c = 0.1 to 0.2 in alcohol) |
| Boiling point | 412.46°C (rough estimate) |
| Density | 1.28±0.1 g/cm3 (20 ºC 760 Torr) |
| refractive index | 210 ° (C=1, EtOH) |
| Flash point | 9℃ |
| storage temp. | -20°C Freezer |
| solubility | Chloroform (Slightly, Heated, Sonicated), DMSO (Slightly), Ethanol (Slightly, Heated) |
| form | Solid |
| pka | 12.37±0.60(Predicted) |
| color | White to Pale Beige |
| Water Solubility | 229.9mg/L(25 ºC) |
| Merck | 2539 |
| Major Application | clinical testing clinical testing |
| InChIKey | MFYSYFVPBJMHGN-ZPOLXVRWSA-N |
| SMILES | O[C@]1([C@@]2([C@H]([C@H]3[C@@H]([C@]4(CCC(=O)C=C4CC3)C)C(=O)C2)CC1)C)C(=O)CO |
| LogP | 1.470 |
| CAS DataBase Reference | 53-06-5(CAS DataBase Reference) |
Safety
| Symbol(GHS) |
  
|
| Signal word | Danger |
| Hazard statements | H225-H301+H311+H331-H370 |
| Precautionary statements | P210-P260-P280-P301+P310-P311 |
| Hazard Codes | F,T,Xn |
| Risk Statements | 11-23/24/25-39/23/24/25-63 |
| Safety Statements | 22-24/25-45-36/37-16-7 |
| RIDADR | UN1230 - class 3 - PG 2 - Methanol, solution |
| WGK Germany | 3 |
| RTECS | GM9020000 |
| F | 18 |
| HS Code | 2937210000 |
| Storage Class | 3 - Flammable liquids |
| Hazard Classifications | Acute Tox. 3 Dermal Acute Tox. 3 Inhalation Acute Tox. 3 Oral Flam. Liq. 2 STOT SE 1 |