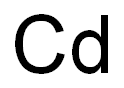Description
Cadmium is a grey-white, soft, blue-white malleable, lustrous metal. It is insoluble in cold water, hot water, methanol, diethyl ether, and n-octanol. It is stable and incompatible with strong oxidising agents, nitrates, nitric acid, selenium, and zinc, and the powdered metal may be pyrophoric and flammable.

Cadmium is associated with occupations such as industrial processes, metal plating, and production of nickel– cadmium batteries, pigments, plastics, and other synthetics. Cadmium metal is produced as a by-product from the extraction, smelting, and refining of the non-ferrous metals zinc, lead, and copper. In view of the unique properties, cadmium metal and cadmium compounds are used as pigments, stabilisers, coatings, specialty alloys, and electronic compounds.
Chemical Properties
Elemental cadmium has a hexagonal crystal structure. Cadmium is a silver-white metal, malleable and ductile, but at 80 C becomes brittle. It remains lustrous in dry air and is only slightly tarnished by air or H2O at standard conditions. The element may be sublimed in a vacuum at a temperature of about 300 C, and when heated in air burns to form the oxide. Cadmium dissolves slowly in hot dilute HCl or H2SO4 and more readily in HNO3. Naturally occurring isotopes 106, 108, 110 114, 116. 113Cd is unstable with respect to beta decay (0.3 MeV) into 113In (t1/2 1013 years).
In virtually all of its compounds, cadmium exhibits the +2 oxidation state, although compounds of cadmium(I) containing the ionCd2 2+, have occasionally been reported. Cadmium hydroxide is more basic than zinc hydroxide, and only slightly amphiprotic, requiring very strong alkali to dissolve it, and forming Cd(OH)3 or Cd(OH)4 2 depending upon the pH.
Chemical Properties
Calmium is an odorless, silver-white lustrous metal with a bluish tinge, which is ductile and highly malleable with a melting point of 321 °C. The metal is soft enough to cut with a knife and will tarnish in air; as a powder, cadmium is flammable. Burning cadmium results in an odorless yellowbrown cadmium fume (cadmium monoxide or cadmium oxide fume) composed of finely divided particles dispersed in air. Both cadmium and cadmium oxide are insoluble in water and have a vapor pressure of approximately 0 mmHg. Cadmium is insoluble in water but can be solubilized in acid. Cadmium salts (e.g., cadmium sulfate and cadmium chloride) are soluble in water.
Physical properties
Cadmium is a soft, blue-white metal that is malleable and ductile although it becomesbrittle at about 80°C. It is also found as a grayish-white powder. It is considered rare and isseldom found by itself as an ore. Its melting point at 320.9°C is considered low. Its boilingpoint is 765°C, and its density is 8.65 g/cm
3. Certain alloys of cadmium have extremely lowmelting points at about 70°C.
Isotopes
There are 52 isotopes of cadmium. Forty-four are radioactive and artificiallyproduced, ranging from Cd-96 to Cd-131. Of these 52 isotopes, there are five stableisotopes plus three naturally occurring radioactive isotopes with extremely long half-livesthat are considered as contributing to the element’s natural occurrence in the Earth’scrust. The three naturally radioactive isotopes (Cd-106, Cd-113, and Cd-116) are thelongest known beta emitters. They are two million years older than when the solar systemwas formed about 4.5 billion years ago. The five stable isotopes and their proportionalcontributions to the element’s existence on Earth are as follows: Cd-108 = 0.89%,Cd-110 = 12.49%, Cd-111= 12.80%, Cd-112 = 24.13%, and Cd-114 = 28.73%.
Origin of Name
The word cadmium is from the Latin word cadmia or the Greek word
kadmeia, meaning the zinc oxide ore “calamine” that contains the element cadmium.
Occurrence
Cadmium is considered a rare element even though it is widely distributed over the Earth’scrust. Its estimated abundance in the Earth’s crust is 1.10
-1 milligrams per kilogram. It is consideredthe 65th most abundant element, but it does not occur as a free metal in nature. It isusually found in relationship with other metallic ores. Its abundance is only about 1/1000ththat of zinc. It is found in an ore called greenockite, which is cadmium sulfite (CdS). This oredoes not have a high enough concentration of cadmium to be mined profitably. Cadmiumis found along with zinc, lead, and copper ores. Today, most cadmium is obtained as a byproductfrom the processing and refining of zinc ores. In addition, dust and fumes from roastingzinc ores are collected by an electrostatic precipitator and mixed with carbon (coke) andsodium or zinc chloride. This residue is then treated to recover the cadmium. Other refiningprocesses can obtain up to 40% recovery of cadmium from zinc ores.
Greenockite ore, as well as zinc and other ores, which produce cadmium as a by-product,are found in many countries, including Australia, Mexico, Peru, Zaire, Canada, Korea, andBelgium-Luxembourg and in the central and western United States.
Characteristics
Although cadmium is not considered a transition element in some periodic tables, it is thecentral element of the triad with zinc and mercury. Zinc is just above it and mercury is below itin group 12 of the periodic table. Cadmium’s chemical and physical properties are similar to itsgroup 12 mates. Their electronegativity is very similar: Zn = 1.6, Cd = 1.7, and Hg = 1.9.
Cadmium is resistant to alkalis, but is soluble in acids, mainly nitric acid. Although it isused to electroplate steel to prevent corrosion, it will tarnish in moist air.
History
Cadmium was discovered by Stromeyer in 1817 from an impurity in zinc carbonate.
Cadmium most often occurs in small quantities associated with
zinc ores, such as sphalerite (ZnS). Greenockite (CdS) is the
only mineral of any consequence bearing cadmium. Almost all
cadmium is obtained as a by-product in the treatment of zinc,
copper, and lead ores. Cadmium is a soft, bluish-white metal which is
easily cut with a knife. It is similar in many respects to zinc. It
is a component of some of the lowest melting alloys; it is used
in bearing alloys with low coefficients of friction and great
resistance to fatigue; it is used extensively in electroplating,
which accounts for about 60% of its use. Cadmium is also used in many
types of solder, for standard E.M.F. cells, for Ni-Cd batteries,
and as a barrier to control atomic fission. The market for Ni-
Cd batteries is expected to grow significantly. Cadmium compounds
are used in black and white television phosphors and
in blue and green phosphors for color TV tubes. It forms a
number of salts, of which the sulfate is most common; the sulfide
is used as a yellow pigment. Cadmium and solutions of its
compounds are toxic. Failure to appreciate the toxic properties
of cadmium may cause workers to be unwittingly exposed
to dangerous fumes. Some silver solders, for example, contain
cadmium and should be handled with care. Serious toxicity
problems have been found from long-term exposure and work
with cadmium plating baths. Cadmium is present in certain
phosphate rocks. This has raised concerns that the long-term
use of certain phosphate fertilizers might pose a health hazard
from levels of cadmium that might enter the food chain. In
1927 the International Conference on Weights and Measures
redefined the meter in terms of the wavelength of the red cadmium
spectral line (i.e., 1 m = 1,553,164.13 wavelengths). This
definition has been changed (see under Krypton). The current
price of cadmium is about 50¢/g (99.5%). It is available in
high purity form for about $550/kg. Natural cadmium is made
of eight isotopes. Thirty-four other isotopes and isomers are
now known and recognized.
Uses
Batteries, including Ni-Cd storage batteries; coating and electroplating steel and cast iron; pigments; plastic stabilizers; constituent of low melting or easily fusible alloys, e.g., Lichtenberg's, Abel's, Lipowitz', Newton's, and Wood's metal; electronics and optics; soft solder and solder for aluminum; reactor control rods; hardener for copper; catalytsts.
Uses
cadmium is highly toxic elements. Cadmium can often be found in batteries.cadmium form the corresponding oxide when heated under oxygen. cadmium follow the general formula MX
2 and are either insoluble (X=F) in water or show a low aqueous solubility.
Uses
Cadmium alloyed with silver forms a type of solder with a low melting point. It is used tojoin electrical junctions and other specialized metallic components. Precautions are required since it is a toxic substance. (Note: This is not the same as common solder used to join metals,which is relatively safe.) Other cadmium alloys are used to manufacture long-wearing bearingsand as thin coatings for steel to prevent corrosion.
Cadmium is a neutron absorber, making it useful as control rods in nuclear reactors. Therods are raised to activate the reactor and then lowered into the reactor to absorb neutronsthat halt the fission reaction.Cadmium, along with nickel, forms a nickel-cadmium alloy used to manufacture “nicadbatteries” that are shaped the same as regular small dry-cell batteries.
.
Uses
Cadmium is used in electroplating, in nickelcadmiumstorage batteries, as a coating forother metals, in bearing and low-meltingalloys, and as control rods in nuclear reactors.Cadmium compounds have numerousapplications, including dyeing and printingtextiles, as TV phosphors, as pigments andenamels, and in semiconductors and solarcells.
Vegetables and cereals are the main sourcesof dietary Cd while meat and fish contain themetal to a lesser extent.
.
Uses
A soft bluish metal, cadmium is extremely toxic, particularly in the compounds used for photography. It is found in zinc ores and in the mineral greenockite (CdS).
Preparation
Cadmium often produces together with Zinc (Zn). The first output part is gathered at the distillation, using the lower boiling point of Cd than that of Zn. Redistillation purifies the material. The Czochralski and horizontal Stockbarger methods are used to grow a single crystal, after melting has taken place.
Thin films are deposited by vacuum evaporation using direct heating in a conical, basket-type heater made of chromel, Nb, Ta, W, Ni, or Fe, or in a Mo or Ta boat. External heating of an alumina crucible with a W heater is also possible. It is also possible to heat an iron crucible externally using a nichrome heater. The evaporation rate at 264 ℃ (sublimation) is 2.67×10
-4 g/cm
2 s.
Production Methods
Two major processes are used for producing cadmium: (1) pyro-hydro-metallurgical and (2) electrolytic. Zinc blende is roasted to eliminate sulfur and to produce a zinc oxide calcine. The latter is the starting material for both processes. In the pyro-hydro-metallurgical process, the zinc oxide calcine is mixed with coal, pelletized, and sintered. This procedure removes volatile elements such as lead, arsenic, and the desired cadmium. From 92 94% of the cadmium is removed in this manner, the vapors being condensed and collected in an electrostatic precipitator. The fumes are leached in H2SO4 to which iron sulfate is added to control the arsenic content. The slurry then is oxidized, normally with sodium chlorate, after which it is neutralized with zinc oxide and filtered. The cake goes to a lead smelter, while the filtrate is charged with highpurity zinc dust to form zinc sulfate or zinc carbonate and cadmium sponge. The latter is briquetted to remove excess H2O and melted under caustic to remove any zinc. The molten metal then is treated with zinc ammonium chloride to remove thallium, after which it is cast into various cadmium metal shapes. The process just described is known as the melting under caustic process. In a distillation process, regular rather than high-purity zinc is used to make the sponge. Then, after washing and centrifuging to remove excess H2O, the sponge is charged to a retort. The heating and distillation process is under a reducing atmosphere. Lead and zinc present in the vapors contaminate about the last 15% of the distillate. Thus, a redistillation is required. The cadmium vapors produced are collected and handled as previously described.
Definition
cadmium: Symbol Cd. A soft bluishmetal belonging to group 12 (formerlyIIB) of the periodic table; a.n.48; r.a.m. 112.41; r.d. 8.65; m.p.320.9°C; b.p. 765°C. The element’sname is derived from the ancientname for calamine, zinc carbonateZnCO
3, and it is usually found associatedwith zinc ores, such as sphalerite(ZnS), but does occur as themineral greenockite (CdS). Cadmiumis usually produced as an associateproduct when zinc, copper, and leadores are reduced. Cadmium is used inlow-melting-point alloys to make solders,in Ni–Cd batteries, in bearingalloys, and in electroplating (over50%). Cadmium compounds are usedas phosphorescent coatings in TVtubes. Cadmium and its compoundsare extremely toxic at low concentrations;great care is essential wheresolders are used or where fumes areemitted. It has similar chemical propertiesto zinc but shows a greater tendencytowards complex formation.The element was discovered in 1817by F. Stromeyer.
General Description
Silver-white blue tinged lustrous metallic solid.
Air & Water Reactions
The finely divided metal is pyrophoric. Slowly oxidized by moist air to form CADMIUM oxide. Insoluble in water.
Reactivity Profile
A violent explosion occurred 30 minutes after placement of a CADMIUM rod into hydrazoic acid [Mellor 8 Supp. 2:50 1967]. Fused ammonium nitrate with powdered metal often produces a violent explosive reaction. Reactivity similar to zinc. May be incompatible with oxidants.
Hazard
Cadmium powder, dust, and fumes are all flammable and toxic if inhaled or ingested.Cadmium and many of its compounds are carcinogenic.
Severe illness and death can occur from exposure to many cadmium compounds. It isabsorbed in the gastrointestinal tract. However, it can be eliminated in the urine and fecesin young, healthy people.
Health Hazard
There are several reports of cadmium poisoningand human death. Cadmium can enterthe body by inhalation of its dusts or fumes,or by ingestion. In humans the acute toxicsymptoms are nausea, vomiting, diarrhea,headache, abdominal pain, muscular ache,salivation, and shock. In addition, inhalationof its fumes or dusts can cause cough,tightness of chest, respiratory distress, congestionin lungs, and bronchopneumonia. A30-minute exposure to about 50 mg/m
3 of itsfumes or dusts can be fatal to humans. Theoral LD
50 value in rats is within the range of250 mg/kg.
Cadmium is a poison that is accumulatedin the liver and kidneys. Thus, chronicpoisoning leads to liver and kidney damage.It is very slowly excreted. Its biologicalhalf-life in humans is estimated atabout 20–30 years (Manahan 1989). Cadmiumlevel in the kidney at 200 μg/g,can damage proximal tubules, resulting intheir inability to reabsorb small-moleculeproteins, such as β
2-microglobulin (Luand Kacey 2003). Cigarette smoking andcalcium-deficient diet enhance its toxicity.Renal toxicity may occur in human subjectsas a result of chronic ingestion of low-leveldietary Cd. The absorption of this metal,however, through the GI tract is low. Cadmiumis also known to produce the so-calleditai-itai disease, which is a chronic renaldisease, producing bone deformity and kidneymalfunction. Cadmium, similar to otherheavy meals, binds to the sulfhydryl (-SH)groups in enzymes, thus inhibiting enzymaticacitivity. Intramuscular administrationof cadmium produced tumors in the lungs and blood in rats. There is sufficient evidenceof its carcinogenicity in animals.
Data on accumulation of Cd in human kidneyand liver have been reviewed by Sataruget al. (2000). Epidemiological and autopsystudies have shown a relationship betweenthe tubular dysfunction and kidney Cd burden.A maximum tolerable level of kidneyCd has been suggested as 50 μg/g (wetweight) corresponding to a urinary excretionof 2 μg Cd per day. Safe daily levels of Cdintake in humans have been recommended tobe kept below 30 μg per day.
Fire Hazard
Flammable in powder form. Combustible.
reaction suitability
core: cadmium
reagent type: catalyst
Industrial uses
Cadmium (symbol Cd) is a silvery-white crystallinemetal that has a specific gravity of 8.6,is very ductile, and can be rolled or beaten intothin sheets. It resembles tin and gives the samecharacteristic cry when bent, but is harder thantin. A small addition of zinc makes it very brittle.It melts at 320°C and boils at 765°C. Cadmiumis employed as an alloying element insoft solders and in fusible alloys, for hardeningcopper, as a white corrosion-resistant platingmetal, and in its compounds for pigments andchemicals. It is also used for Ni–Cd batteriesand to shield against neutrons in atomic equipment;but gamma rays are emitted when theneutrons are absorbed, and these rays requirean additional shielding of lead.
Most of the consumption of cadmium is forelectroplating. For a corrosion-resistant coatingfor iron or steel a cadmium plate of 0.008 mmis equal in effect to a zinc coat of 0.025 mm.The plated metal has a silvery-white color witha bluish tinge, is denser than zinc, and harderthan tin, but electroplated coatings are subjectto H2 embrittlement, and aircraft parts are usuallycoated by the vacuum process. Cadmiumplating is not normally used on copper or brasssince copper is electronegative to it, but whenthese metals are employed next to cadmium-plated steel a plate of cadmium may beused on the copper to lessen deterioration.
Safety Profile
Confirmed human
carcinogen with experimental carcinogenic,
tumorigenic, and neoplastigenic data. A
human poison by inhalation and possibly
other routes. Poison experimentally by
ingestion, inhalation, intraperitoneal,
Potential Exposure
Cadmium is a highly corrosion resistant and is used as a protective coating for iron, steel, and
copper; it is generally applied by electroplating, but hot
dipping and spraying are possible. Cadmium may be
alloyed with copper, nickel, gold, silver, bismuth, and aluminum to form easily fusible compounds. These alloys
may be used as coatings for other materials; welding electrodes, solders, etc. It is also utilized in electrodes of alkaline storage batteries, as a neutron absorber in nuclear
reactors, a stabilizer for polyvinyl chloride plastics, a deoxidizer in nickel plating; an amalgam in dentistry; in the
manufacture of fluorescent lamps, semiconductors,photocells, and jewelry, in process engraving, in the automobile and aircraft industries; and to charge Jones
reductors. Various cadmium compounds find use as fungicides, insecticides, nematocides, polymerization catalysts,
pigments, paints, and glass; they are used in the photographic industry and in glazes. Cadmium is also a contaminant of superphosphate fertilizers. Human exposure to
cadmium and certain cadmium compounds occurs through
inhalation and ingestion. The entire population is exposed
to low levels of cadmium in the diet because of the entry
of cadmium into the food chain as a result of its natural
occurrence. Tobacco smokers are exposed to an estimated
17 μg/cigarette. Cadmium is present in relatively low
amounts in the earth’s crust; as a component of zinc ores,
cadmium may be released into the environment around
smelters
First aid
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. Give large quantities of water and inducevomiting. Do not make an unconscious person vomit.Medical observation is recommended for 24-48 h afterbreathing overexposure, as pulmonary edema may bedelayed. As first aid for pulmonary edema, a doctor orauthorized paramedic may consider administering a corticosteroid spray.Note to physician: For severe poisoning do not use BAL[British Anti-Lewisite, dimercaprol, dithiopropanol(C3H8OS2)] as it is contraindicated or ineffective inpoisoning from cadmium.
Carcinogenicity
Cadmium and cadmium compounds are known to be human carcinogens based on sufficient evidence of carcinogenicity from studies in humans, including epidemiological and mechanistic studies. Cadmium and cadmium compounds were first listed as reasonably anticipated to be human carcinogens in the First Annual Report on Carcinogens in 1980, based on sufficient evidence of carcinogenicity from studies in experimental animals. The listing was revised to known to be human carcinogens in the Ninth Report on Carcinogens in 2000.
Environmental Fate
Cadmium inhibits plasma membrane calcium channels and
Ca2t-ATPases. It also inhibits repair of DNA damaged by
various chemicals, an effect which is believed to be associated
with the induction of tumors. Although cadmium forms
a metallothionein, the preformed cadmium metallothionein is
nephrotoxic (toxic to the kidneys); it is suggested that effects
occur when, at some stage in the kidney, the cadmium is
dissociated from the metallothionein. In Itai-Itai disease (see
Human under Chronic Toxicity section), patients were found
to have chromosome abnormalities.
Cadmium has an affinity for sulfhydryl groups, and hence
can inhibit enzymes; however, cells treated with cadmium
showed proliferation of peroxisomes, which contain catalase,
an enzyme. It appears that cadmium at first inhibits catalase
activity and then, after a time, enhances that activity. In addition,
cadmium inhibits enzymes involved in gluconeogenesis
(the generation of glycogen for energy production from noncarbohydrate
precursors). It also inhibits oxidative phosphorylation
(energy production) and depresses trypsin inhibitor
capacity.
storage
Cadmium should be kept stored in a tightly closed container in a cool place. It should be
kept stored in a separate locked safety storage cabinet
Shipping
UN2570 Cadmium compounds, Hazard Class:
6.1; Labels: 6.1-Poisonous materials, Technical Name
Required.
Purification Methods
Any oxide contaminant is removed by filtering the molten metal, under vacuum, through quartz wool. Its solubility in Hg is 5.2% (18o), and it is soluble in mineral acids. [Wagenknecht & Juza in Handbook of Preparative Inorganic Chemistry (Ed. Brauer) Academic Press Vol II p 1092 1965.]
Structure and conformation
The space lattice of Cadmium belongs to the hexagonal system, and its closely-packed hexagonal lattice has lattice constants of a=0.2973 nm and c=0.5607 nm.
Toxicity evaluation
As indicated in the Exposure and Exposure Monitoring section,
cadmium is widely distributed in the environment from
a variety of natural and anthropogenic sources. Cadmium
emitted into the air is often found bound to small particulates
and can travel with these particulates over long distances. As
a result, cadmium can remain in the atmosphere for long
periods of time until it is deposited by gravitational settling or
in rain and snow. Cadmium tends to be more mobile in water
than other heavy metals although it will complex with humic
substances and can precipitate out under certain conditions.
Cadmium can bioaccumulate in aquatic organisms; the degree
of accumulation is associated with the pH and humic content
of the water. It can also bioaccumulate in plants and in the
animals that feed on these plants; for example, cattle and
wildlife. However, terrestrial bioaccumulation is much lower
than that in water and cadmium concentrations at the top of
the terrestrial food chain are not much higher than those at the
lower end of the chain.
Incompatibilities
Air exposure with cadmium powder may
cause self-ignition. Moist air slowly oxidizes cadmium
forming cadmium oxide. Cadmium dust is incompatible
with strong oxidizers, ammonium nitrate; elemental sulfur;
hydrazoic acid; selenium, zinc, tellurium. Contact with
acids cause a violent reaction, forming flammable hydrogen
gas.
Toxics Screening Level
The initial risk screening level (IRSL) for cadmium is 0.0006 μg/m3 (annual averaging time) based on the Environmental Protection Agency’s (EPA’s) inhalation unit risk, 0.0018 per μg/m3 (EPA 1992, MDNR 1992).
Waste Disposal
With cadmium compounds in
general, precipitation from solution as sulfides, drying and
return of the material to suppliers for recovery is recommended. Cadmium may be recovered from battery scrap as
an alternative to disposal. In accordance with
40CFR165, follow recommendations for the disposal of
pesticides and pesticide containers. Must be disposed properly by following package label directions or by contacting
your local or federal environmental control agency, or by
contacting your regional EPA office.
Precautions
On exposures to cadmium, wash the skin immediately with plenty of water and a nonabrasive soap. Workers should cover the exposed skin with an emollient.