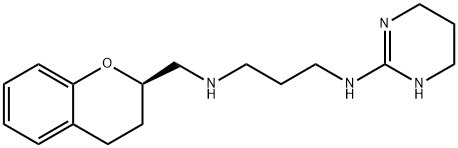
Alniditan
Bezeichnung:Alniditan
CAS-Nr152317-89-0
Englisch Name:Alniditan
CBNumberCB43175523
SummenformelC17H26N4O
Molgewicht302.41
MOL-Datei152317-89-0.mol
Alniditan physikalisch-chemischer Eigenschaften
| storage temp. | Store at -20°C |
| Löslichkeit | Soluble in DMSO |
Alniditan Chemische Eigenschaften,Einsatz,Produktion Methoden
-
Originator
Pasmigren ,Janssen-Cilag -
Verwenden
Antimigraine. -
Manufacturing Process
To a stirred and heated +80°C mixture of 3,4-dihydro-2H-1-benzopyran-2- carboxylic acid and methylbenzene were added dropwise thionyl chloride during a period of 85 min. Upon complete addition, stirring was continued for 2 h at 80°C. After cooling to room temperature, the reaction mixture was evaporated. The residue was taken up in methylbenzene and the solvent was evaporated again, yielding (R)-3,4-dihydro-2H-1-benzopyran-2-carbonyl chloride.
A mixture of (R)-3,4-dihydro-2H-1-benzopyran-2-carbonyl chloride in N,Ndimethylacetamide and 2,2'-oxybispropane was hydrogenated in the presence of palladium-on-charcoal catalyst (10%) and a solution of thiophene in methanol (4%). After the calculated amount of hydrogen was taken up, the catalyst was filtered off and the filtrate was added to a mixture of benzenemethanamine, potassium acetate and methanol. This mixture was hydrogenated again in the presence of palladium-on-charcoal catalyst (10%) and a solution of thiophene in methanol (4%). After the calculated amount of hydrogen was taken up, the catalyst was filtered off and the filtrate was evaporated. The residue was poured into water and the whole was basified with NaOH (50%). The product was extracted with dichloromethane and the extract was dried, filtered and evaporated. The residue was purified by column chromatography (silica gel; CH2Cl2/CH3OH 95:5). The eluent of the desired fraction was evaporated and the residue was converted into the hydrochloride salt in 2-propanone by adding 2-propanol saturated with HCl. The salt was filtered off and dried, yielding (R)-3,4-dihydro-N-(phenylmethyl)-2H-1- benzopyran-2-methanamine.
A mixture of 28.0 g of (R)-3,4-dihydro-N-(phenylmethyl)-2H-1-benzopyran-2- methanamine and 300 ml of methanol was hydrogenated in the presence of 2.0 g of palladium-on-charcoal catalyst (10%). After the calculated amount of hydrogen was taken up, the catalyst was filtered off and the filtrate was evaporated, yielding 18.2 g (100%) of (-)-(R)-3,4-dihydro-2H-1-benzopyran- 2-methanamine as crude residue.
A mixture of 18.0 g of (-)-(R)-3,4-dihydro-2H-1-benzopyran-2-methanamine, 60.0 g of 2-propenenitrile and 400 ml of ethanol was stirred for 4 h at reflux temperature. The reaction mixture was evaporated and the residue was dried, yielding 20.0 g (84%) of (-)-(R)-3-[[(3,4-dihydro-2H-1-benzopyran-2- yl)methyl]amino]propanenitrile.
A mixture of 20.0 g (-)-(R)-3-[[(3,4-dihydro-2H-1-benzopyran-2- yl)methyl]amino]propanenitrile and 300 ml of methanol was hydrogenated in the presence of 5.0 g of Raney Nickel. After the calculated amount of hydrogen was taken up, the catalyst was filtered off and the filtrate was evaporated, yielding 21.0 g (100%) of (-)-(R)-N-[(3,4-dihydro-2H-1- benzopyran-2-yl)methyl]-1,3-propanediamine as crude residue.
A mixture of (-)-(R)-N-[(3,4-dihydro-2H-1-benzopyran-2-yl)methyl]-1,3- propanediamine, 2-chloropyrimidine, sodium carbonate and ethanol was stirred for 4 h at reflux temperature. The reaction mixture was evaporated. The residue was purified by column chromatography (silica gel; CHCl3/CH3OH 90:10). The eluent of the desired fraction was evaporated and the residue was converted into the hydrochloride salt in 2-propanol. The salt was filtered off and dried in vacuum, yielding (-)-(R)-N-[(3,4-dihydro-2H-1-benzopyran-2- yl)methyl]-N'-(2-pyrimidinyl)-1,3-propanediamine dihydrochloride hemihydrate. A mixture of 3.6 g of (-)-(R)-N-[(3,4-dihydro-2H-1-benzopyran-2-yl)methyl]- N'-(2-pyrimidinyl)-1,3-propanediamine dihydrochloride hemihydrate in 150 ml of methanol and 20 ml of 2-propanol saturated with HCl was hydrogenated in the presence of 1.5 g of palladium-on-charcoal catalyst (2%). After the calculated amount of hydrogen was taken up, the catalyst was filtered off and the filtrate was evaporated. The product was crystallized from acetonitrile, filtered off and dried, yielding 2.7 g (74.0%) of (-)-(R)-N-[(3,4-dihydro-2H-1- benzopyran-2-yl)methyl]-N'-(1,4,5,6-tetrahydro-2- -
Therapeutic Function
Migraine therapy -
in vitro
In vitro, alniditan exhibits little vasoconstrictive effects on the rat basilar artery, although at a very high concentration 1 mM, alniditan causes intensive constriction, most likely through a mechanism independent from 5-HT receptor activation. Alniditan is 10 times more potent than sumatriptan at the h5-HT1B receptor, and twice as potent at the h5-HT 1D receptor. -
in vivo
The intraperitoneal administration of alniditan ED 50 =9 μg/kg and sumatriptan ED 50 =70 μg kg dose dependly reduces [ 125 I]-BSA extravasation in the rat meninges when done 30 min before stimulation. The estimated ED values for alniditan are 9 μg/kg in the absence and 190 μg/kg in the presence of GR 127935. Alniditan (3, 10, 30 and 100 μg/kg) produces a dose-dependent increase in the arteriovenous oxygen saturation difference, which seems to be attenuated in animals treated with GR127935. Alniditan dose-dependently decreases total carotid and arteriovenous anastomotic blood flow and concomitant conductance values; nutrient blood flow and conductance increase. Alniditan also produces significant increases in vascular conductance to the skin, ear, bone, salivary gland, fat, tongue, brain and dura mater; no changes are observed in the muscles and eyes.
Alniditan Anbieter Lieferant Produzent Hersteller Vertrieb Händler.
Global(11)Suppliers
-
Telefon +1-781-999-5354<br/>+1-00000000000
E-Mail marketing@targetmol.com
-
Telefon
E-Mail support@targetmol.com
-
Telefon +8613564774135
E-Mail zijue.cai@tsbiochem.com
-
Fan De(Beijing) Biotechnology Co., Ltd.
Telefon 15911056312
E-Mail liming@bio-fount.com
-
Beijing xinyanhui pharmaceutical research and development co., LTD
Telefon 13969155946
E-Mail 1461866103@qq.com
-
Telefon 13348960310<br/>13348960310
E-Mail 3003867561@qq.com
-
Beijing Jin Ming Biotechnology Co., Ltd.
Telefon 010-60605840<br/>15801484223;
E-Mail psaitong@jm-bio.com
-
Telefon 4008200310
E-Mail marketing@tsbiochem.com
-
Suzhou Zhixin Biotechnology Co., Ltd.
Telefon 0512-65118909<br/>15162312715
E-Mail sales@szzxbio.com
-
Nantong QuanYi Biotechnology Co., Ltd
Telefon 0513-66337626<br/>18051384581
E-Mail sales@chemhifuture.com
1of2
