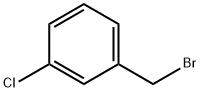
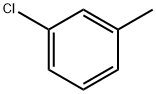
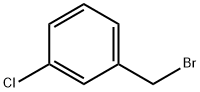
General procedure for the synthesis of 3-chlorobenzyl bromide from 3-chlorotoluene: In a reaction flask fitted with a stirrer, reflux condenser, and thermometer, m-chlorotoluene (5.1 g, 40 mmol), sodium bromate (2.1 g, 14 mmol), sodium bromide (2.9 g, 28 mmol), and dichloromethane (25 mL) were added. A tail gas absorption device was installed and heated to reflux. AIBN (0.025 g) and BPO (0.025 g) were dissolved in 5 mL of dichloromethane and quickly added to the reaction flask. Subsequently, diluted concentrated sulfuric acid (2.1 g, 21 mmol, diluted with 2.5 mL of water) was added slowly and dropwise. Upon completion of the reaction, the progress of the reaction was monitored by gas chromatography. The mixture was cooled to room temperature, saturated sodium bisulfite solution (10 mL) was added and stirred until the solution faded from red to colorless. The aqueous phase was extracted twice with dichloromethane (10 mL x 2), the organic phases were combined, washed with saturated sodium chloride solution, dried over anhydrous sodium sulfate and concentrated under reduced pressure. The crude product was purified by column chromatography (eluent was petroleum ether) to give 6.8 g of 3-chlorobenzyl bromide as a colorless liquid in 83% yield.