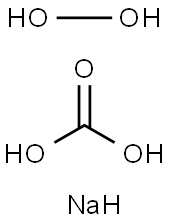
Перкарбонат натрия
- английское имяSodium percarbonate
- CAS №15630-89-4
- CBNumberCB4853147
- ФормулаCH5NaO5
- мольный вес120.04
- EINECS239-707-6
- номер MDLMFCD00036142
- файл Mol15630-89-4.mol
| плотность | 2.09[at 20℃] |
| давление пара | < 10-3 Pa at 25°C |
| форма | Granular Powder |
| цвет | White |
| Запах | Odorless |
| РН | About 10.5 at 1% concentration (20°C) |
| Растворимость в воде | Soluble in water. |
| Чувствительный | Moisture Sensitive |
| Разложение | > 131 °F (> 55 °C) |
| LogP | -0.809 (est) |
| Справочник по базе данных CAS | 15630-89-4(CAS DataBase Reference) |
| Рейтинг продуктов питания EWG | 1-2 |
| FDA UNII | Z7G82NV92P |
| Система регистрации веществ EPA | Sodium percarbonate (15630-89-4) |
| UNSPSC Code | 12352302 |
| NACRES | NA.23 |
| Коды опасности | O,Xn | |||||||||
| Заявления о рисках | 8-22-41-36/38 | |||||||||
| Заявления о безопасности | 17-26-39-37/39 | |||||||||
| РИДАДР | UN 1479 5.1/PG 3 | |||||||||
| WGK Германия | 1 | |||||||||
| RTECS | FG0750000 | |||||||||
| F | 21 | |||||||||
| TSCA | Yes | |||||||||
| Класс опасности | 5.1 | |||||||||
| Группа упаковки | II | |||||||||
| кода HS | 28369990 | |||||||||
| Банк данных об опасных веществах | 15630-89-4(Hazardous Substances Data) | |||||||||
| Токсичность | mouse,LD50,oral,2200mg/kg (2200mg/kg),BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY)LUNGS, THORAX, OR RESPIRATION: DYSPNEA,Toksikologicheskii Vestnik. Vol. (3), Pg. 46, 1994. | |||||||||
| NFPA 704: |
|
рисовальное письмо(GHS)
-
рисовальное письмо(GHS)



-
сигнальный язык
опасность
-
вредная бумага
H302:Вредно при проглатывании.
H318:При попадании в глаза вызывает необратимые последствия.
H272:Окислитель; может усилить возгорание.
-
оператор предупредительных мер
P210:Беречь от тепла, горячих поверхностей, искр, открытого огня и других источников воспламенения. Не курить.
P220:Не допускать соприкосновения с одеждой и другими горючими материалами.
P264:После работы тщательно вымыть кожу.
P280:Использовать перчатки/ средства защиты глаз/ лица.
P301+P312:ПРИ ПРОГЛАТЫВАНИИ: Обратиться за медицинской помощью при плохом самочувствии.
P305+P351+P338:ПРИ ПОПАДАНИИ В ГЛАЗА: Осторожно промыть глаза водой в течение нескольких минут. Снять контактные линзы, если Вы ими пользуетесь и если это легко сделать. Продолжить промывание глаз.
Перкарбонат натрия химические свойства, назначение, производство
Химические свойства
Sodium percarbonate is a white granular powder of sodium carbonate and hydrogen peroxide.
Sodium percarbonate is mainly used as a bleaching chemical in laundry detergents (tablets, compact or regular powders), laundry additives and machine dishwashing products. It is an oxidizing agent and ingredient in a number of home and laundry cleaning products, including eco-friendly bleach products such as OxiClean and Tide laundry detergent. Dissolved in water, it releases hydrogen peroxide and soda ash (sodium carbonate):
2(Na2CO31.5H202)→2Na2CO3+3H202
Использование
Sodium percarbonate is an addition salt of hydrogen peroxide and sodium carbonate that provides a solid source of hydrogen peroxide. When dissolved in water, sodium percarbonate liberates hydrogen peroxide. Sodium percarbonate is a white, granular or powdered solid oxidizer. It is used primarily as a bleaching agent in cleaning products. Other uses include algaecides, fungicides, chemical synthesis and environmental applications such as control of odor at waste treatment facilities. A small amount is used in denture cleaners and toothpaste.Multifunctional reagent for the preparation of optically active 4-hydroxy-2-isoxazolines.
Подготовка
Sodium percarbonate is produced by the reaction of sodium carbonate with hydrogen peroxide, which can be done via dry, spray and wet processes. In the dry process aqueous hydrogen peroxide solution is sprayed on solid sodium carbonate; a solid-liquid reaction yields sodium percarbonate. In the spray process sodium percarbonate is produced by a fluid bed process. Solutions of sodium carbonate and hydrogen peroxide are sprayed into a drying chamber where the water is evaporated. In the wet process sodium percarbonate is usually prepared by cristallisation possibly in combination with salting out.Реакции
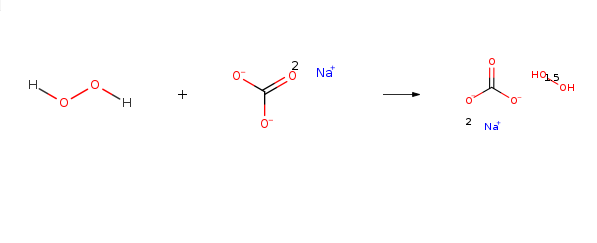
Sodium percarbonate naturally decomposes, very slowly, to form sodium carbonate and hydrogen peroxide. The hydrogen peroxide may further decompose to form water and oxygen and liberate some heat. The decomposition proceeds according to the reaction below:2Na2CO3 • 3H2O2 → 2Na2CO3 + 3H2O + 1.5 O2 + Heat
Общее описание
A colorless, crystalline solid. Denser than water. May combust in contact with organic materials. Contact may irritate skin, eyes and mucous membranes. May be toxic by ingestion. Used to make other chemicals.Реакции воздуха и воды
Soluble in water.Профиль реактивности
Oxidizing agents, such as SODIUM PERCARBONATE, can react with reducing agents to generate heat and products that may be gaseous (causing pressurization of closed containers). The products may themselves be capable of further reactions (such as combustion in the air). The chemical reduction of materials in this group can be rapid or even explosive, but often requires initiation (heat, spark, catalyst, addition of a solvent). Explosive mixtures of inorganic oxidizing agents with reducing agents often persist unchanged for long periods if initiation is prevented. Such systems are typically mixtures of solids, but may involve any combination of physical states. Some inorganic oxidizing agents are salts of metals that are soluble in water; dissolution dilutes but does not nullify the oxidizing power of such materials. Organic compounds, in general, have some reducing power and can in principle react with compounds in this class. Actual reactivity varies greatly with the identity of the organic compound. Inorganic oxidizing agents can react violently with active metals, cyanides, esters, and thiocyanates.Угроза здоровью
Inhalation, ingestion or contact (skin, eyes) with vapors or substance may cause severe injury, burns or death. Fire may produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may cause pollution.Пожароопасность
These substances will accelerate burning when involved in a fire. Some may decompose explosively when heated or involved in a fire. May explode from heat or contamination. Some will react explosively with hydrocarbons (fuels). May ignite combustibles (wood, paper, oil, clothing, etc.). Containers may explode when heated. Runoff may create fire or explosion hazard.Профиль безопасности
Moderately toxic by ingestion. When heated to decomposition it emits acrid smoke and irritating vaporsСинтез
Sodium percarbonate is prepared by the reaction of dihydrogen peroxide and sodium carbonate. The specific synthesis steps are as follows:With florisil In water addn. of small amounts of MgSiO3 and water glass to 500kg H2O2 (dild. to 20% soln.), addn. of 1051kg Na2CO3 at 18°C, cooling to 5°C, cooling to -4°C after addn. of 500kg NaCl; centrifugation, drying, reuse of mother-liquor, finally sodium percarbonate (yield: 85.5%) is obtained.
Экологическая судьба
Sodium percarbonate is not persistent in the environment and readily decomposes to soda ash (sodium carbonate) and hydrogen peroxide which will subsequently decompose to water and oxygen when exposed to soils, sediments, and surface or ground waters.Перкарбонат натрия запасные части и сырье
сырьё
1of2
запасной предмет
- 3-(6-FORMYL-PYRIDIN-2-YL)-BENZONITRILE
- 6-(3-NITROPHENYL)-2-PYRIDINECARBOXALDEHYDE
- 6-(4-METHYLSULFANYLPHENYL)PYRIDINE-2-CARBALDEHYDE
- 6-(4-METHOXYPHENYL)PYRIDINE-2-CARBALDEHYDE
- 6-(THIOPHEN-3-YL)PYRIDINE-2-CARBALDEHYDE
1of3
Перкарбонат натрия поставщик
| поставщик | телефон | страна | номенклатура продукции | благоприятные условия |
|---|---|---|---|---|
| +86-13131129325 | China | 5887 | 58 | |
| +8619931165850 | China | 1000 | 58 | |
| +86-17736087130 +86-18633844644 |
China | 994 | 58 | |
| +86-0371-55170693 +86-19937530512 |
China | 21632 | 55 | |
| +86-0551-65418679 +8618949832763 |
China | 2986 | 55 | |
| +86-0371-86658258 +8613203830695 |
China | 29871 | 58 | |
| +86 18953170293 | China | 2930 | 58 | |
| 18871490254 | CHINA | 28172 | 58 | |
| +86-86-5926051114 +8618959220845 |
China | 6383 | 58 | |
| 86-13657291602 | CHINA | 22963 | 58 |