N,N-Diethylhydroxylamine: Applications in Enhancing Photosystem II Electron Transport and its Health Hazards
General Description
N,N-Diethylhydroxylamine has emerged as a significant compound in enhancing electron transport within photosystem II (PSII) in plant thylakoid membranes, supporting crucial processes like dichlorophenolindophenol (DCPIP) photoreduction and electron transport with methyl viologen. Its unique ability to donate electrons without inactivating the oxygen-evolving complex sets it apart from other electron donors, maintaining PSII function and ensuring consistent benzoquinone Hill activity. This stability under varying light conditions highlights its effectiveness and potential in photosynthetic research, offering new insights into PSII mechanisms and applications in agriculture and environmental sciences. However, N,N-Diethylhydroxylamine poses several health hazards, including respiratory and skin irritation, organ weight changes, and potential carcinogenicity. Chronic exposure has shown severe effects like increased stomach tumors in female mice and significant health impacts in inhalation studies with rats. Therefore, handling this compound with stringent safety measures is imperative to mitigate associated health risks.

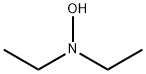
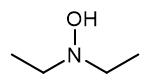
Figure 1. N,N-Diethylhydroxylamine
Applications in Enhancing Photosystem II Electron Transport
Unique Electron Donor Capabilities
N,N-Diethylhydroxylamine has emerged as a pivotal compound in enhancing electron transport within photosystem II (PSII) in plant thylakoid membranes. This compound exhibits notable effects when introduced into reaction mixtures involving PSII-mediated processes such as dichlorophenolindophenol (DCPIP) photoreduction and electron transport supported by methyl viologen. Studies have demonstrated that N,N-diethylhydroxylamine effectively supports DCPIP photoreduction even after the inactivation of the oxygen-evolving complex through hydroxylamine washings. This indicates its role in donating electrons to PSII, particularly prior to the binding site for herbicides like diuron, which significantly affect electron transport assays. Importantly, N,N-diethylhydroxylamine's impact on photochemical activity is not solely attributable to its uncoupling properties. Unlike other electron donors like hydroxylamine, N,N-diethylhydroxylamine does not necessitate the inactivation of the oxygen-evolving complex to facilitate electron donation. This distinguishes it as a unique electron donor capable of maintaining PSII function without compromising benzoquinone Hill activity in thylakoid membranes.
Stability and Application Potential
Furthermore, experiments have shown that pre-incubations with N,N-diethylhydroxylamine, both in light and darkness, do not alter its electron-donating capabilities, reinforcing its stability and effectiveness under varying conditions of light exposure. This stability and specificity in electron donation make N,N-diethylhydroxylamine a valuable tool in studying and enhancing PSII-mediated processes in photosynthesis research. In conclusion, N,N-diethylhydroxylamine stands out as a potent electron donor to photosystem II, facilitating critical biochemical reactions without disrupting essential PSII functions. Its application opens avenues for further understanding PSII electron transport mechanisms and exploring novel approaches in agricultural and environmental sciences reliant on efficient photosynthetic processes. 1
Health Hazards
Respiratory and Dermatological Irritation
N,N-Diethylhydroxylamine poses various health hazards. N,N-Diethylhydroxylamine is classified as an upper respiratory tract irritant, causing mild eye and skin irritation in rabbits. Inhalation studies in rats have shown decreased body weights, hypoactivity, changes in white blood cell counts, reduced thymus weight, and increased liver weight after exposure to N,N-Diethylhydroxylamine. Additionally, exposure to N,N-Diethylhydroxylamine led to transient changes in the nasal mucosa of rats. In a chronic exposure study in mice, treatment with N,N-Diethylhydroxylamine resulted in a significant increase in stomach tumors in females.
Carcinogenic Potential and Systemic Effects
Furthermore, in a developmental toxicity study in rats, maternal animals exhibited reduced weight gain and food consumption when exposed to N,N-Diethylhydroxylamine during gestation. However, no developmental toxicity was observed in the offspring. N,N-Diethylhydroxylamine reacts exothermically with water, acts as an eye and respiratory tract irritant, and can reduce available oxygen for respiration when inhaled. In a case report, N,N-Diethylhydroxylamine was detected in the urine of a man who ingested a silicone resin precursor containing N,N-Diethylhydroxylamine, leading to fatal consequences. Overall, the toxic effects of N,N-Diethylhydroxylamine include irritations, changes in organ weights, decreased body weights, and potential carcinogenicity in certain cases. It is crucial to handle N,N-Diethylhydroxylamine with care and ensure proper safety measures are in place to prevent exposure and minimize health risks associated with this chemical compound. 2
References:
[1] S.C. SABAT P M T Sudhakar Babu. N,N-diethylhydroxylamine: A new electron donor to photosystem II[J]. Biochemical and biophysical research communications, 1991, 179 2: 713-1140. DOI:10.1016/0006-291X(91)91937-8.You may like
Related articles And Qustion
See also
Lastest Price from N,N-Diethylhydroxylamine manufacturers

US $150.00/kg2025-04-21
- CAS:
- 3710-84-7
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 500kg

US $85.00-25.00/kg2025-04-15
- CAS:
- 3710-84-7
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 20ton