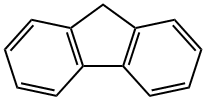
Fluorene: Degradation and Biological Hazard
Fluorene is a polycyclic aromatic hydrocarbon used in various applications such as polymers, electronic devices, sensors, and photochromic materials. This white crystalline solid is characterized by its distinctive structure and unique chemical properties.

Synthesis of Fluorene
In advance, Cs2CO3 was dried by heating under vacuum for 3 h then stored in a glovebox. A 10-mL Schlenk flask was charged with the dried Cs2CO3 (0.16 g, 0.50 mmol) in a glovebox, then taken out of glovebox and dried by heating under a vacuum for at least 5 min. The flask was backfilled with argon and then PdCl2(PPh3)2 (1.4 mg, 0.0020 mmol, 1.0 mol%) and pivalic acid (1c; 20 mg, 0.20 mmol, 1.0 equiv) were added. The flask was then evacuated and backfilled with argon three times. THF (0.50 mL) and 2a (37 L, 0.20 mmol) were added, and the mixture was stirred at 25 °C for 18 h. The mixture was then analyzed by GC with tetradecane (50 L) as an internal standard. H2O (5 mL) and EtOAc (5 mL) were added, and the mixture was extracted with EtOAc (3 × 5 mL). The collected organic layers were combined, washed with brine (5 mL), dried (MgSO4), and concentrated. The residue (Fluorene) was purified by chromatography (silica gel, hexane-acetone) to give a colorless solid; yield: 32.1 mg (96%). 1H NMR (400 MHz, CDCl3): δ = 7.80 (d, J = 7.8 Hz, 2 H), 7.56 (d,J = 7.3 Hz, 2 H), 7.39 (t, J = 7.3 Hz, 2 H), 7.31 (td, J = 7.4, 1.1 Hz, 2H), 3.92 (s, 2 H). 13C NMR (100 MHz, CDCl3): = 143.2, 141.7, 126.7, 126.7, 125.0, 119.8, 36.9. EI-HRMS: m/z: [M - H]+ calcd for C13H9: 165.0704; found: 165.0702.[1]
Evidence for a novel pathway in the degradation of fluorene
A fluorene-utilizing microorganism, identified as a species of Pseudomonas, was isolated from soil severely contaminated from creosote use and was shown to accumulate six major metabolites from fluorene in washed-cell incubations. Five of these products were identified as 9-fluorenol, 9-fluorenone, (+)-1,1a-dihydroxy-1-hydro-9-fluorenone, 8-hydroxy-3,4-benzocoumarin, and phthalic acid. This last compound was also identified in growing cultures supported by fluorene. Fluorene assimilation into cell biomass was estimated to be approximately 50%. The structures of accumulated products indicate that a previously undescribed pathway of fluorene catabolism is employed by Pseudomonas sp. strain F274. This pathway involves oxygenation of fluorene at C-9 to give 9-fluorenol, which is then dehydrogenated to the corresponding ketone, 9-fluorenone. Dioxygenase attack on 9-fluorenone adjacent to the carbonyl group gives an angular diol, 1,1a-dihydroxy-1-hydro-9-fluorenone. Identification of 8-hydroxy-3,4-benzocoumarin and phthalic acid suggests that the five-membered ring of the angular diol is opened first and that the resulting 2'-carboxy derivative of 2,3-dihydroxy-biphenyl is catabolized by reactions analogous to those of biphenyl degradation, leading to the formation of phthalic acid. Cell extracts of fluorene-grown cells possessed high levels of an enzyme characteristic of phthalate catabolism, 4,5-dihydroxyphthalate decarboxylase, together with protocatechuate 4,5-dioxygenase. On the basis of these findings, a pathway of fluorene degradation is proposed to account for its conversion to intermediary metabolites. A range of compounds with structures similar to that of fluorene was acted on by fluorene-grown cells to give products consistent with the initial reactions proposed.[2]
Bioactive fluorenes
In this study, a new series of 2,7-dichloro-4-(2-substituted-amino acetyl)fluorene derivatives were synthesized, characterized and evaluated for their antimicrobial activity and screened for cytotoxic activity against human lung carcinoma (A-549) and human breast carcinoma (MCF-7) cell lines. Fluorene and its derivatives are versatile reagents which are used in wide range of synthetic applications. Fluorene-based aromatic ketones are of profound interest as building blocks for the production of drugs, pharmaceuticals and industrial fine chemicals particularly in the production of lubricating and thermosetting plastic materials. Furthermore, fluorene-based polymers and copolymers are of interest owing to their unusual optical and electrical properties and therefore are usually used in organic light-emitting diodes, flat panel displays and in solar cells.[3]
Benflumetol is a racemic fluorene derivative, which mimics the structure and reactivity of the arylamino alcohol group of antimalarial drugs such as quinine, mefloquine, and halofantrine and is used as antimalarial agent in combination treatment with artemether. In the synthesis of benflumetol, 2,7-dichloro-4-(chloroacetyl)fluorene is an important intermediate Based on these previous findings, we envisaged that the hybridization of the versatile 2,7-dichlorofluorene moiety with α-N-aryl-, N-alkyl, N-dialkylamino acetyl pharmacophores into a single chemical entity could be effective in enhancing its anticancer and antimicrobial properties. To achieve this, our previous experience in the synthesis of new antimicrobial and anticancer agents was helpful. In the present work, we have synthesized some new 2,7-dichloro-4-(2-substitutedamino acetyl)fluorenes and studied their cytotoxic activity against human lung carcinoma (A-549) and human breast carcinoma (MCF-7) cell lines followed by antimicrobial and molecular docking studies.
Scientists report herein the synthesis of a new series of 2,7-dichloro-4-(2-substituted-amino acetyl)fluorene derivatives. Most of these new compounds exhibited significant anticancer activity against human lung carcinoma (A-549) and human breast carcinoma (MCF-7) cell lines, when compared to 5-Fluorouracil as a reference drug. In addition, on the antimicrobial evaluation; some of these synthesized compounds showed acceptable activity as antibacterial and antifungal agents. To the best of our knowledge, these multi-addressable properties of the new synthesized 2,7-dichlorofluorene derivatives described in this work will open a new alternative era in the field of medicinal chemistry and can be considered as pharmacophores.
Fluorene exposure is associated with epigenetic markers related to lung cancer
Exposure to high-molecular-weight polycyclic aromatic hydrocarbons (PAHs) may cause cancer in chimney sweeps and creosote-exposed workers, however, knowledge about exposure to low-molecular-weight PAHs in relation to cancer risk is limited. In this study, we aimed to investigate occupational exposure to the low-molecular-weight PAHs phenanthrene and fluorene in relation to different cancer biomarkers. Exposure to high-molecular-weight polycyclic aromatic hydrocarbons (PAHs) may cause cancer in chimney sweeps and creosote-exposed workers, however, knowledge about exposure to low-molecular-weight PAHs in relation to cancer risk is limited. In this study, we aimed to investigate occupational exposure to the low-molecular-weight PAHs phenanthrene and fluorene in relation to different cancer biomarkers.[4]
Despite the frequent presence of fluorene in PAH mixtures, only a limited number of studies have explored its toxicity. An in vitro study using Chinese hamster lung cell line reported a clastogenic effect of fluorene only in the presence of metabolic activation (rat S9 mix). Fluorene also showed axial development toxicity in zebrafish embryos and sea urchin embryos through dysregulation of the β-catenin/Wnt signalling pathway. An in ovo study assessing histopathological changes in the chicken fetal liver on exposure to various compounds found no genotoxicity, measured as DNA adducts, of fluorene. Due to the scarcity of toxicological studies, and thus, the inadequate evidence of fluorene carcinogenicity, IARC has classified fluorene in group 3.
We recruited 151 chimney sweeps, 19 creosote-exposed workers and 152 unexposed workers (controls), all men. We measured monohydroxylated metabolites of phenanthrene and fluorene in urine using liquid chromatography coupled to tandem mass spectrometry. We measured, in peripheral blood, the cancer biomarkers telomere length and mitochondrial DNA copy number using quantitative PCR; and DNA methylation of F2RL3 and AHRR using pyrosequencing.Chimney sweeps and creosote-exposed workers are occupationally exposed to phenanthrene and fluorene. We report for the first time that fluorene exposure is associated with the DNA hypomethylation of F2RL3 and AHRR, prospective markers for lung cancer, in the chimney sweeps. Further studies are needed to clarify whether fluorene or other PAHs/toxicants are underlying the epigenetic alterations observed in workers exposed to PAHs.
References
[1]Tanji, Yutaka; Tsuji, Yasushi; Fujihara, Tetsuaki[Synlett, 2020, vol. 31, # 8, p. 805 - 808]
[2]Grifoll M, Selifonov SA, Chapman PJ. Evidence for a novel pathway in the degradation of fluorene by Pseudomonas sp. strain F274. Appl Environ Microbiol. 1994 Jul;60(7):2438-49.
[3]Hussein EM, Alsantali RI, Abd El-Galil SM, Obaid RJ, Alharbi A, Abourehab MAS, Ahmed SA. Bioactive fluorenes. part I. Synthesis, pharmacological study and molecular docking of novel dihydrofolate reductase inhibitors based-2,7-dichlorofluorene. Heliyon. 2019 Jun 26;5(6):e01982.
[4]Alhamdow A, Essig YJ, Krais AM, Gustavsson P, Tinnerberg H, Lindh CH, Hagberg J, Graff P, Albin M, Broberg K. Fluorene exposure among PAH-exposed workers is associated with epigenetic markers related to lung cancer. Occup Environ Med. 2020 Jul;77(7):488-495.
You may like
Lastest Price from Fluorene manufacturers

US $10.00/ASSAYS2025-08-21
- CAS:
- 86-73-7
- Min. Order:
- 1ASSAYS
- Purity:
- 99%
- Supply Ability:
- 1 ton

US $10.00/KG2025-04-21
- CAS:
- 86-73-7
- Min. Order:
- 100KG
- Purity:
- 99%
- Supply Ability:
- 100 mt