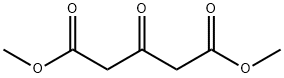
Dimethyl 1,3-acetonedicarboxylate: a highly reactive compound used in organic synthesis
Dimethyl 1,3-acetonedicarboxylate is a highly reactive compound used in organic synthesis for diverse applications, such as UV protection and pharmaceutical development. This kind of substance is a colorless liquid.

Synthesis of Dimethyl 1,3-acetonedicarboxylate
Methanol absorbs hydrogen chloride: 100.0 g of sodium chloride and 150 mL of concentrated sulfuric acid are sequentially added to a 500 mL three-necked flask, and concentrated sulfuric acid is slowly added dropwise while stirring. The generated hydrogen chloride gas is passed to a concentrated sulfuric acid reagent bottle to dry, and then passed to In the two-necked flask, the other bottle was connected to a drying tube, and the gas at the outlet of the drying tube was checked with a pH test paper. When the test paper became red, the hydrogen chloride methanol solution reached saturation.423 g of methanol (199 g of hydrogen chloride dissolved) was transferred into a flask equipped with a calcium chloride drying tube, and 65 g (0.445 mol) of 1,3-acetone dicarboxylic acid was added. The water bath was heated to 36 ° C, and after reacting for 3.5 hours, cooled to room temperature. Firstly add 776 mL of water, separate the ester layer, and then add 689 mL of dichloromethane to take the organic phase. The aqueous phase was back-extracted with 242 mL of dichloromethane. After the organic phases were combined, the organic phase was washed with an aqueous sodium bicarbonate solution to pH = 7.0, and then washed twice with saturated brine, and dried over anhydrous magnesium sulfate. Concentrated at atmospheric pressure and vacuum rectified to obtain the product of Dimethyl 1,3-acetonedicarboxylate.[1]
Reactions of 3-acylchromones with dimethyl 1,3-acetonedicarboxylate and 1,3-diphenylacetone
Reactions of 3-methoxalyl-, 3-polyfluoroacyl- and 3-aroylchromones with dimethyl 1,3-acetonedicarboxylate and 1,3-diphenylacetone in the presence of DBU proceed at the C-2 atom of the chromone system with pyrone ring-opening and subsequent formal [3 + 3] cyclocondensation to functionalized 2-hydroxybenzophenones, 6H-benzo[c]chromenes and benzo[c]coumarins, depending on the substituent at the 3-position. An NMR study and X-ray crystallographic analysis are reported. The compounds synthesized can be considered as promising scaffolds for the design of the novel UV-A/B and UV-B filters.
3-Formylchromones are also known to undergo Michael addition and cyclization reactions with dimethyl 1,3-acetonedicarboxylate under basic conditions to produce similarly functionalized 2-hydroxybenzophenones. 3-substituted chromones are widely used as valuable synthetic intermediates in the preparation of new heterocycles, examples of the participation of chromones1–3 in any reactions with 1,3-C,C-dinucleophiles are lacking, a fact prompting us to investigate their reactions with dimethyl 1,3-acetonedicarboxylate and 1,3-diphenylacetone. Continuing our research program dedicated to the design and synthesis of novel heterocyclic compounds and in view of the unique biological properties displayed by many chromones and coumarin derivatives Iaroshenko's laboratory started investigation in this area by the study of the reactions of chromones1–3 with dimethyl 1,3-acetonedicarboxylate.[2]
In conclusion, scientists have developed a simple and convenient method for the synthesis of functionalized 2-hydroxybenzophenones, 6H-benzo[c]chromenes and benzo[c]coumarins by formal [3 + 3] cyclocondensations of 3-acylchromones with dimethyl 1,3-acetonedicarboxylate and 1,3-diphenylacetone. The products constitute an important structural subunit of a variety of biologically active compounds, which are not readily available by other methods. The biological evaluation of the synthesized compounds is currently being studied in our laboratories. Nevertheless, due to the strong UV-absorption properties of many of the representatives the libraries synthesized here can also find a use in the preparation of the sun-protective materials and creams.
Reaction between alkyl isocyanides and dimethyl 1,3-acetonedicarbocxylate
The reaction of alkyl isocyanides with dimethyl 1,3-acetonedicarboxylate in the presence of dialkyl acetylenedicarboxylates in CH2Cl2 at ambient temperature leads to highly functionalized 2-amino-4H-pyrans and 1,2-dialkyl 4,6-dimethyl-(1E, 3E)-3 (alkylamino)-5-oxo-1,3-hexadiene-1,2,4,6-tetracarboxylates. We previously reported the reaction between alkyl isocyanides and cyclic 1,3-diketones in the presence of acetylenic esters [13]. In the current work, we wish to report that a one-pot chemo and stereoselective reaction between alkyl isocyanides 1 and dialkyl acetylenedicarboxylates 2 in the presence of dimethyl 1,3-acetonedicarboxylate 3 can produce the desired highly functionalized 2-amino-4H-pyran derivatives 4a–4f and enamino carbonyl compounds 5a–5f.[3]
Although we have not yet established the mechanism of the reaction between alkyl isocyanides and acetylenic esters in the presence of dimethyl 1,3-acetonedicarboxylate in an experimental manner. In conclusion, the chemo- and stereoselective three com ponent reactions of alkyl isocyanides with electron deficient acetylenic esters in the presence of dimethyl 1,3-acetonedi carboxylate provides a simple one-pot entry in to the syn thesis of highly poly functional 4H-pyran derivatives and enaminocarbonyl compounds of potential synthetic interest. The present method carries the advantage that not only is the reaction performed under natural condition but also the starting material and reagents can be mixed without any activation or modification.
References
[1]LANXI LISHUN BIOLOGY - CN110407866, 2019, A
[2]Iaroshenko, V. O., Savych, I., Villinger, A., Sosnovskikh, V. Ya., & Langer, P. (2012). Reactions of 3 - acylchromones with dimethyl 1,3 - acetonedicarboxylate and 1,3 - diphenylacetone: one - pot synthesis of functionalized 2 - hydroxybenzophenones, 6 H - benzo[c]chromenes and benzo[c]coumarins. Organic Biomolecular Chemistry, 47.
[3]Nasiri F, Nazem F, Pourdavaie K. Chemo- and stereoselective reaction between alkyl isocyanides and dimethyl 1,3-acetonedicarbocxylate in the presence of acetylenic esters. Mol Divers. 2007 May;11(2):101-5.
You may like
Related articles And Qustion
See also
Lastest Price from Dimethyl 1,3-acetonedicarboxylate manufacturers

US $0.00-0.00/kg2025-04-21
- CAS:
- 1830-54-2
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 20MT

US $30.00-10.00/KG2025-04-15
- CAS:
- 1830-54-2
- Min. Order:
- 50KG
- Purity:
- 99%
- Supply Ability:
- 500000kg