A useful reagent: Trifluoromethanesulfonic anhydride
Description
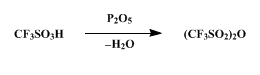
Trifluoromethanesulfonic anhydride ((CF3SO2)2O; Tf2O) has proved to be a versatile reagent in organic synthesis over the last few decades[1]. Due to the excellent leaving group properties of the triflate group, Tf2O finds its use in converting various compounds to triflates, which can be transformed into other valuable products.
Uses
Trifluoromethanesulfonic anhydride is employed as an electrophilic reagent for the conversion of various oxygen-containing compounds to the triflates, such as carbonyl compounds, alcohols, and phenols, while the triflate group is one of the best-leaving groups used in nucleophilic substitution reactions and cross-coupling reactions. Oxygen-containing nucleophiles, such as sulfoxides and phosphorus oxides, could also undergo nucleophilic attack with Tf2O to generate thionium triflate, electrophilic P-species, and phosphonium triflate[2]. These highly active transient species could readily undergo nucleophilic substitution reactions for further diverse transformations. Furthermore, owing to the strong electrophilic property, Tf2O is prone to react with relatively weak nucleophiles such as nitrile group or some nitrogen-containing heterocyclic compounds, and a series of exciting transformations based on this strategy have been developed during the past several years.
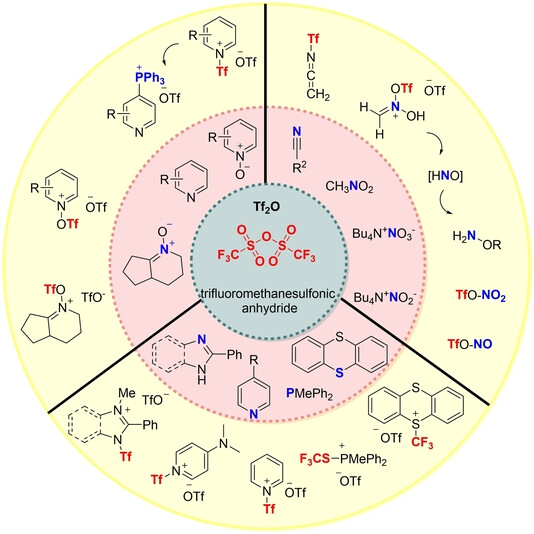
Nitrogen-containing heterocycles, such as pyridines, pyridine N-oxides, pyrimidines, benzothiazoles, and 2H-azirines could be readily activated by Trifluoromethanesulfonic anhydride and followed by nucleophilic substitution with various reagents to afford the corresponding products. Similar to the electrophilic activation of amides, sulfoxides, and phosphorus oxides, the activation of the nitro group with Tf2O would result in a variety of new transformations, either as a nitrogen donor to form amides and nitriles or as nitro and nitroso precursor.
It is an inexpensive and abundant chemical and thus represents an attractive and practical trifluoromethylation reagent. Yao et al. report the unprecedented direct trifluoromethylation of (hetero)arenes using trifluoromethanesulfonic anhydride as a radical trifluoromethylation reagent by merging photoredox catalysis and pyridine activation[3]. Furthermore, introducing CF3 and OTf groups of trifluoromethanesulfonic anhydride into internal alkynes to access tetrasubstituted trifluoromethylated alkenes is developed.
References
[1] Sarkar, S. “Trifluoromethanesulfonic (triflic) Anhydride.” Synlett 108 1 (2004): 576–576.
[2] Dr. Qixue Qin, Prof.?Dr. Ning Jiao, Zengrui Cheng. “Recent Applications of Trifluoromethanesulfonic Anhydride in Organic Synthesis.” Angewandte Chemie International Edition 62 10 (2022).
[3] Yao Ouyang, Feng-Ling Qing, Xiu-Hua Xu. “Trifluoromethanesulfonic Anhydride as a Low-Cost and Versatile Trifluoromethylation Reagent.” Angewandte Chemie International Edition 57 23 (2018): 6926–6929.
You may like
Related articles And Qustion
Lastest Price from Trifluoromethanesulfonic anhydride manufacturers

US $0.00-0.00/KG2025-05-15
- CAS:
- 358-23-6
- Min. Order:
- 1KG
- Purity:
- 98.0%
- Supply Ability:
- 10000KGS

US $0.00/G/KG2025-04-22
- CAS:
- 358-23-6
- Min. Order:
- 1G/KG
- Purity:
- 99%
- Supply Ability:
- 100MT