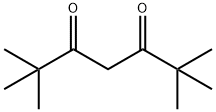
2,2,6,6-Tetramethyl-3,5-heptanedione: conformational properties and metal chelates
2,2,6,6-Tetramethyl-3,5-heptanedioneis a stable, anhydrous reagent. It undergoes O-additions and C-additions. In various reactions, it acts as an air-stable ligand for metal catalysts. Furthermore, it serves as a substrate for heterocycles. 2,2,6,6-tetramethyl-3,5-heptanedione was used in the synthesis of α-aryl-β-diketones and dicyanamidobenzene-bridge diruthenium complex.

Synthesis of 2,2,6,6-Tetramethyl-3,5-heptanedione
Step a: Feeding: The mixture of Methyl pivalate, sodium ethoxide and formamide is mixed and stirred for 2.5 hours to obtain the mixture. The molar ratio of methyl pivalate to base is 1: 3.Step b: Reaction: The mixture in step a is continuously stirred under heating at 55 ° C, and the ketone is slowly added thereto, and the reaction is carried out at atmospheric pressure for 8-48 h to obtain a reaction solution containing the product; And the frequency of the ketone ketone is 1: 1;Step c: Purification: Water is added to the reaction solution of step b, and the mixture is stirred uniformly. The reaction solution is washed several times with ethyl acetate. The washed reaction solution is adjusted to pH with nitric acid and extracted with organic solvent. Organic phase, concentrated, vacuum distillation. The final product 2,2,6,6-Tetramethyl-3,5-heptanedione is obtained.[1]
Tautomeric and conformational properties of 2,2,6,6-Tetramethyl-3,5-heptanedione
The tautomeric and structural properties of 2,2,6,6-Tetramethyl-3,5-heptanedione, (dipivaloylmethane, C(CH3)3C(O)CH2C(O)C(CH3)3) have been studied by means of gas-phase electron diffraction (GED) and quantum chemical calculations (B3LYP and MP2 approximation with different basis sets up to aug-cc-pVTZ). Both, quantum chemistry and GED analyses resulted in the presence of 100(5)% enol tautomer at 296(3)K. Quantum chemical calculations predict the existence of two enol conformers in about equal amounts. In both conformers the enol ring possesses Cs symmetry and they possess different torsional orientations of the two tert-butyl groups. In the literature it has been reported that the substitution of methyl groups in acac by more bulky groups (CF3, phenyl or t-butyl) should increase the enol content. The larger the alkyl substituent, the greater are steric interactions, resulting in carbonyl groups to be closer to each other, and hence the tendency to form the enol tautomer increases. Furthermore, D.C. Nonhebel has suggested that the strong electron donating effect of two t-butyl groups favors the intramolecular hydrogen bond in the enol form of 2,2,6,6-Tetramethyl-3,5-heptanedione (dpm) as compared to that in acac. In the present study we are interested in the effect of the substitution of methyl by more bulky t-butyl groups on the tautomeric and conformational properties by investigating 2,2,6,6-tetramethyl-3,5-heptanedione, dipivaloylmethane (C(CH3)3C(O)CH2C(O)C(CH3)3).[2]
Two series of GED/MS experiments at two different nozzle-to-plate distances were performed. The conditions of GED/MS experiments and the relative abundance of the characteristic ions of 2,2,6,6-Tetramethyl-3,5-heptanedione are shown, respectively. A special inlet system constructed for studying liquids was used for inserting vapor to the effusion cell. The temperature of the stainless steel effusion cell was measured with a W/Re-5/20 thermocouple calibrated by the melting points of Sn and Al. A least squares refinements with 5–7% contribution of a diketo tautomer lead to a increase of Rf. Using Hamilton's criteria with 0.01 significance level we obtain a tautomeric composition of 100(5)% enol. Thus, the re-interpretation of GED experimental data confirms the quantum chemical calculations which predict an asymmetric enol ring structure of dipivaloylmethane with a localized OH bond, independent of the computational method.
Fractional sublimation of various metal chelates of 2,2,6,6-Tetramethyl-3,5-heptanedione
Twenty different metal chelates of 2,2,6,6-Tetramethyl-3,5-heptanedione were prepared and characterized by melting points, carbon-hydrogen analyses, and infrared and n.m.r. absorption data. The pure chelates were sublimed in the Berg fractional vacuum sublimator and the temperature condensation (sublimation) zones were recorded for each chelate under standardized conditions. The beryllium(II) chelate was the most volatile and the lanthanide and actinide chelates among the least volatile 2,2,6,6-Tetramethyl-3,5-heptanedione. The only volatile alkaline earth metal chelates were those of Be(II) and Mg(II); the Ca(II), Sr(II) and Ba(II) chelates were not volatile under the conditions used. Overall it was found that the chelates of 2,2,6,6-Tetramethyl-3,5-heptanedione exhibited volatilities similar to those of the corresponding metal acetylacetonates but less than those of the trifluoroacetylacetonates and the hexafluoroacetylacetonates. The chelates of benzoylacetone, benzoyltrifluoroacetone and thenoyltrifluoroacetone are less volatile than the corresponding chelates of dipivaloylmethane. The data suggest that some 64 different binary and ternary mixtures of metal 2,2,6,6-Tetramethyl-3,5-heptanedione chelates can be quantitatively resolved. Seven binary and three ternary mixtures of the metal chelates were resolved satisfactorily.[3]
References
[1]HARVARD UNIVERSITY - WO2019/9956, 2019, A1
[2]Belova, N. V., Nguyen, H. T., Oberhammer, H., & Girichev, G. V. (2017). Tautomeric and conformational properties of dipivaloylmethane. Journal of Molecular Structure, 1132, 63-69.
[3]Berg, E. W., & Herrera, N. M. (1972). Fractional sublimation of various metal chelates of dipivaloylmethane. Analytica Chimica Acta, 60(1), 117-125.
See also
Lastest Price from 2,2,6,6-Tetramethyl-3,5-heptanedione manufacturers
US $0.00/kg2025-08-26
- CAS:
- 1118-71-4
- Min. Order:
- 1kg
- Purity:
- 99%min
- Supply Ability:
- 20tons
US $205.00-195.00/KG2025-04-21
- CAS:
- 1118-71-4
- Min. Order:
- 25KG
- Purity:
- 98%
- Supply Ability:
- 500kg