Treatment of malaria, African
sleeping sickness, and pneumocystis pneumonia.
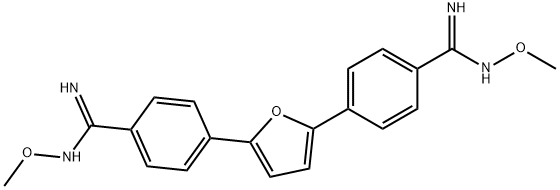
Pafuramidine is a prodrug of Furamidine (F863600, 2HCl); a diphenylfuran compound belonging to an important class of antimicrobial and antiparasitic agents. Furamidine and its analogues also display antitumor activities and showed antiproliferative activities against various tumor cell lines.
pafuramidine, an orally bioavailable prodrug of furamidine (db75) with considerable trypanocidal activity, is an experimental drug for the treatment of pneumocystis pneumonia (pcp). pafuramidine is well tolerated and has clinical activity against pneumocystis pneumonia[1].
inmurine models of human african trypanosomiasis, clearance of parasites from the peripheral circulation started 48 h after initiation of treatment with pafuramidineand was complete in all groups 6 days after the first drug dose. administration of pafuramidine po or ip at dose rates equal to or greater than 4 mg/kg resulted in 100% cure rates [2]. in the vervet monkey (chlorocebus [cercopithecus] aethiops) model of sleeping sickness, pafuramidine (10 mg/kg) completely cured all three monkeys, whereas lower doses of 3 mg/kg and 1 mg/kg cured only one of three and zero of three monkeysin an early-stage infection, respectively. in a late-stage infection, pafuramidine treatment resulted in cure rates of one of three and zero of three monkeys. these data indicated the limited ability of pafuramidine to cross the blood-brain barrier [3].
[1] chen d, marsh r, aberg j a. pafuramidine for pneumocystis jiroveci pneumonia in hiv-infected individuals[j]. expert review of anti-infective therapy, 2007, 5(6): 921-928.
[2] thuita j k, karanja s m, wenzler t, et al. efficacy of the diamidine db75 and its prodrug db289, against murine models of human african trypanosomiasis[j]. actatropica, 2008, 108(1): 6-10.
[3] mdachi r e, thuita j k, kagira j m, et al. efficacy of the novel diamidine compound 2, 5-bis (4-amidinophenyl)-furan-bis-o-methlylamidoxime (pafuramidine, db289) against trypanosoma bruceirhodesiense infection in vervet monkeys after oral administration[j]. antimicrobial agents and chemotherapy, 2009, 53(3): 953-957.
[4] harrilla h, desmet k d, wolf k k, et al. a mouse diversity panel approach reveals the potential for clinical kidney injury due to db289 not predicted by classical rodent models[j]. toxicological sciences, 2012: kfs238.