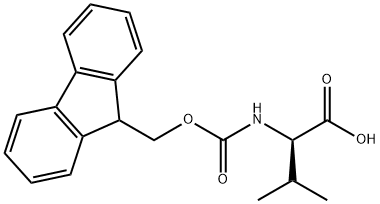
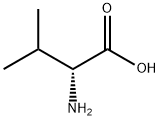
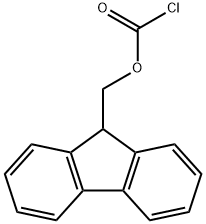
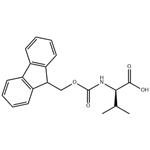
General procedure for the synthesis of Fmoc-D-valine from D-valine and chloroformic acid-9-fluorenylmethyl ester: A 500 mL three-necked flask was taken, and D-valine (4.5 g, 38.9 mmol), dioxane (40 mL), and 10% aqueous sodium carbonate (100 mL) were added sequentially. The reaction system was placed in an ice bath and mechanical stirring was initiated. A dioxane solution (100 mL) of methyl chloroformate-9-fluorenyl (10.0 g, 38.8 mmol) was prepared in a dropping funnel and slowly added dropwise to the reaction vial. After the dropwise addition was completed, it was gradually brought to room temperature and stirring was continued overnight. After completion of the reaction, 100 mL of water was added to the reaction mixture and extracted three times with 50 mL of ether and the organic phase was discarded. The aqueous phase was cooled in an ice bath, and the pH was adjusted to 1 by slowly adding 1 M dilute hydrochloric acid, followed by extracting the aqueous phase with 50 mL ethyl acetate three times, combining the organic phases and drying with anhydrous magnesium sulfate. The desiccant was removed by filtration and the organic phase was concentrated under reduced pressure to give the white solid product Fmoc-D-valine (12.6 g, 96% yield).