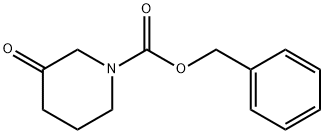
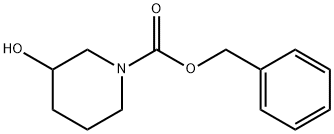
General procedure for the synthesis of 1-N-Cbz-3-piperidone from N-Benzyloxycarbonyl-3-hydroxypiperidine: N-Benzyloxycarbonyl-3-hydroxypiperidine (16.50 g, 70 mmol) was dissolved in dichloromethane (90 mL), stirred and cooled to 0 °C. Subsequently, N,N-diisopropylethylamine (DIPEA, 45.30 g, 0.35 mol) was added to the reaction system. A solution of dimethyl sulfoxide (DMSO, 100 mL) of pyridine trioxide (25.70 g, 0.16 mol) was slowly added dropwise at 0 °C. After the dropwise addition was completed, the reaction mixture was continued to be stirred at 0°C for 2 hours. After completion of the reaction, the mixture was poured into 4 M hydrochloric acid and extracted with dichloromethane (100 mL x 3). The organic layers were combined, washed with saturated brine, dried over anhydrous magnesium sulfate, filtered and concentrated under reduced pressure. The resulting residue was purified by silica gel column chromatography with the eluent ethyl acetate/petroleum ether (1/100 to 1/6, v/v) to afford 1-N-Cbz-3-piperidone (14.90 g, 90% yield) as an off-white solid. The structure of the product was confirmed by LC-MS, LC-MS: [M + H]+ = 234.