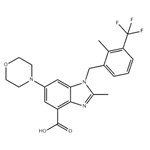
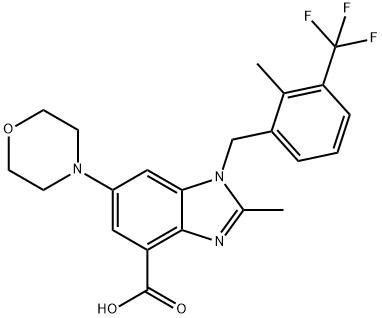
2-Methyl-1-{[2-methyl-3-(trifluoromethyl)phenyl]methyl}-6-(4-morpholinyl)-1H-benzimidazole-4-carboxylate (180 mg, 0.4 mmol) was used as a starting material and dissolved in THF (10 mL). To this solution, 2N LiOH aqueous solution (1.2 mL) was added, followed by stirring the reaction at 50 °C for 1 hour. The progress of the reaction was monitored by TLC and after confirming complete consumption of the feedstock, the reaction mixture was cooled to room temperature. THF was removed under reduced pressure and the mixture was subsequently acidified by adjusting the pH to 3. The resulting suspension was filtered and the filter cake was washed with water (10 mL) to give the final white solid product 2-methyl-1-[[2-methyl-3-(trifluoromethyl)phenyl]methyl]-6-(4-morpholinyl)-1H-benzimidazole-4-carboxylic acid (152 mg, yield). The product was characterized by 1H NMR (300 MHz, DMSO-d6): δ 2.46 (s, 3H), 2.54 (s, 3H), 3.10 (t, 4H, J = 4.8 Hz), 3.73 (t, 4H, J = 4.8 Hz), 5.63 (s, 2H), 6.37 (d, 1H, J = 7.8 Hz), 7.26 (t, 1H, J = 7.8 Hz), 7.35 (d, 1H, J = 2.4 Hz), 7.44 (d, 1H, J = 2.4 Hz), 7.62 (d, 1H, J = 7.8 Hz); LC-MS: m/e = 434 [M + 1]+.
![methyl 2-methyl-1-(2-methyl-3-(trifluoromethyl)benzyl)-6-morpholino-1H-benzo[d]imidazole-4-carboxy](/CAS/20180808/GIF/1372540-24-3.gif)