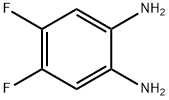
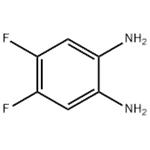
4,5-Difluorobenzene-1,2-diamine is an organic synthesis intermediate and a chemical and pharmaceutical intermediate, which can be used in the research and development of organic synthesis reactions in laboratories and in the research and development of chemical and pharmaceutical products.
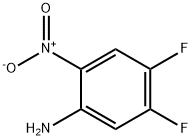
General procedure for the synthesis of 4,5-difluorophthalimide from 2-nitro-4,5-difluoroaniline: 4,5-difluoro-2-nitroaniline (10 g, 57 mmol) was co-suspended in methanol (150 mL) with 10% palladium carbon catalyst (2.0 g, 1.9 mmol). The reaction system was transferred to a hydrogenation unit and the catalytic hydrogenation reaction was carried out under 50 psi hydrogen pressure. After 3 hours of reaction, the catalyst was removed by filtration through a diatomaceous earth pad and the filtrate was concentrated under reduced pressure to give a solid product. The resulting solid was dried under vacuum at room temperature overnight to obtain white crystalline 4,5-difluorophthalimide (8.05 g, 98% yield).
[1] Journal of Medicinal Chemistry, 1993, vol. 36, # 3, p. 331 - 342
[2] Patent: US2005/43292, 2005, A1. Location in patent: Page/Page column 14
[3] Patent: EP2766359, 2016, B1. Location in patent: Paragraph 0755
[4] Journal of Medicinal Chemistry, 1997, vol. 40, # 5, p. 811 - 818
[5] Russian Journal of Organic Chemistry, 2001, vol. 37, # 4, p. 564 - 569