A white or almost white, voluminous powder.
Thephorin ,Roche, US,1947
Histamine H1 receptor antagonist; antihistamine.
A mixture of 750 grams of 1-methyl-3-benzoyl-4-hydroxy-4-phenylpiperidine and 2,500 cc of 48% hydrobromic acid is refluxed for about 20 minutes. It is then poured into 8 liters of water. An oily precipitate appears which on standing crystallizes. It is filtered and crystallized from about 3.5 liters of alcohol. 2-Methyl-9-phenyl-2,3-dihydrel-pyridindene hydrobromide, MP 201°203°C, is obtained.
A mixture of 680 grams of 2-methyl-9-phenyl-2,3-dihydrol-pyridindene hydrobromide, 6,000 cc of water and about 100 grams of Raney-nickel catalyst is hydrogenated at room temperature and at about 1,000 lb pressure for a period of three hours. The catalyst is filtered. The clear filtrate is treated with a solution of 240 grams potassium thiocyanate in 400 cc of water. A heavy solid precipitates from which the supernatant liquid is decanted.
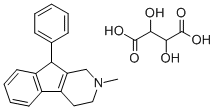
The residue is dissolved in 10 liters of boiling alcohol with stirring in the presence of nitro gen. The solution is cooled to room temperature under nitrogen, and then allowed to stand overnight. 2-Methyl-9-phenyl-tetrahydro1-pyridindene thiocyanate separates in crystals of MP 188°-189°C. From the concentrated filtrate an additional amount is obtained. The corresponding free base, prepared by treating the slightly soluble thiocyanate in aqueous suspension with sodium hydroxide and extracting with ether, has a MP of 90°91°C. It forms a tartrate of MP 160°C.
The starting material was prepared by reacting acetophenone, methylamine and formaldehyde followed by treatment of the intermediate with sodium hydroxide.
Thephorin (Hoffmann-LaRoche).
Phenindamine tartrate,2,3,4,9-tetrahydro-2-methyl-9-phenyl-1H-indeno[2, 1-c]pyridine bitartrate, occurs as a creamy-white powder, usuallywith a faint odor and sparingly soluble in water (1:40). A 2%aqueous solution has a pH of about 3.5. It is most stable inthe pH range of 3.5 to 5.0 and is unstable in solutions of pH 7or higher. Oxidizing substances or heat may cause isomerizationto an inactive form.
Structurally, phenindamine can be regarded as an unsaturatedpropylamine derivative, in that the rigid ring systemcontains a distorted, trans-alkene. Like the other commonlyused antihistamines, it may produce drowsiness and sleepiness;it may also cause a mildly stimulating action in somepatients and insomnia when taken just before bedtime.