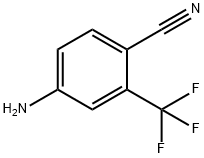
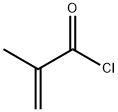
Methacryloyl chloride (2.63 mL, 27.16 mmol) was slowly added dropwise to a stirred N,N-dimethylacetamide (14 mL) solution of 4-amino-2-trifluoromethylbenzonitrile (3.4 mmol) under nitrogen protection for a controlled time period of 10 min and the reaction was carried out at room temperature. The progress of the reaction was monitored by TLC for 3 h or overnight (o.n.). Upon completion of the reaction, the reaction mixture was diluted with ethyl acetate (100 mL) and washed sequentially with saturated aqueous sodium bicarbonate solution (3 x 25 mL) and cold brine (4 x 50 mL). The organic layer was dried over anhydrous sodium sulfate and concentrated under reduced pressure to remove the solvent. The crude product was purified by fast column chromatography to afford N-(4-cyano-3-trifluoromethylphenyl)methacrylamide in white solid form in 95% yield. The structure of the product was confirmed by 1H-NMR (CDCl3): δ 8.06 (d, J = 2.0 Hz, 1H), 7.99 (dd, J = 8.5 Hz, 2.0 Hz, 1H), 7.87 (bs, 1H), 7.82 (d, J = 8.5 Hz, 1H), 5.90-5.88 (m, 1H), 5.63-5.61 (m, 1H). 2.11-2.10 (m, 3H).
![N-[4-Cyano-3-(trifluoromethyl)phenyl]-2-methacrylamide Structure](/CAS/GIF/90357-53-2.gif)

![N-[4-Cyano-3-(trifluoromethyl)phenyl]-2-methacrylamide pictures](/ProductImageEN/2023-06/Small/98ac29b3-eed7-4b1c-9e7f-e7c29a2a05e5.gif)
![N-[4-Cyano-3-(trifluoromethyl)phenyl]-2-methacrylamide pictures](https://img.chemicalbook.com/ProductImageEN/2020-1/Small/27510fc3-6fee-4cc6-87e8-a8294fbab043.png)