Nystatin was found in the mycelium of Streptomyces noursei in 1950 and produced on an industrial scale by Squibb & Sons Co. in 1954. This antibiotic was used orally and topically as the first clinically applied polyene macrolide with antifungal properties. Nystatin shows activity against Candida and filamentous fungi and is used to treat Candida infections of the mouth, digestive organs, and vagina. The application of nystatin in combination with gentamicin and vancomycin to sterilize the gut in perioperation of bone-marrow transplantation has been developed recently.
This substance is a yellow or yellow-brown powder with a unique smell and the ability to absorb moisture. It dissolves to a small extent in water, methanol, and ethanol, but cannot dissolve in acetone, ether, or chloroform. It is not stable when exposed to air, light, heat, water, acid, or alkali.
Mycostatin,Squibb,US,1954
Nystatin is polyene antifungal containing a conjugated tetraene and a diene, isolated as a complex of three components A1, A2 and A3 from Streptomyces noursei and first reported in 1950. Nystatin, like most polyene antifungals, binds to sterols in the fungal cell membrane leading to formation of ion channels in the wall, ion imbalance and cell death. Nystatin is an established bioprobe and widely-used antifungal reagent with over 4,000 literature citations.
Polyene antifungal antibiotic complex containing 3 biologically active components, A1, A2, A3. Increases the permeability of the cell membrane of sensitive fungi by binding to sterols. Cell culture tested. Growth promotant.
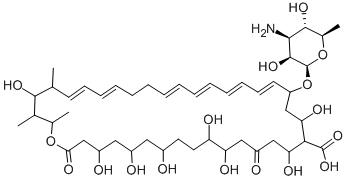
Nystatin A1 is shown.
ChEBI: A polyene macrolide antibiotic; part of the nystatin complex produced by several Streptococcus species. The keto-form of nystatin A1.
Nystatin (Mycostatin) is a polyene antifungal drug with
a ring structure similar to that of amphotericin B and a
mechanism of action identical to that of amphotericin
B. Too toxic for systemic use, nystatin is limited to the
topical treatment of superficial infections caused by
C. albicans. Infections commonly treated by this drug
include oral candidiasis (thrush), mild esophageal candidiasis,
and vaginitis.
A typical isolation and recovery procedure for nystatin is described in US Patent 2,797,183 and is shown in the following diagram:
Barstatin 100 (Barlan); Candex (Bayer); Korostatin (Holland Rantos); Mycostatin (Bristol-Myers Squibb); Mycostatin (Westwood-Squibb).
Nystatin (Mycostatin) is a polyene antibiotic that was first isolated in 1951 from a strain of the actinomycete Streptomyces noursei by Hazen and Brown.33 It occurs as a yellow to light tan powder. Nystatin is very slightly soluble in water and sparingly soluble in organic solvents. The compound is unstable to moisture, heat, and light. The aglycone portion of nystatin is called nystatinolide. The complete structure of nystatin has been determined by chemical degradation and x-ray crystallography.35 Nystatin is not absorbed systemically when administered by the oral route. It is nearly insoluble under all conditions. It is also too toxic to be administered parenterally. Hence, it is used only as a topical agent. Nystatin is a valuable agent for the treatment of local and gastrointestinal monilial infections caused by C. albicans and other Candida species. For the treatment of cutaneous and mucocutaneous candidiasis, it is supplied as a cream, an ointment, and a powder. Vaginal tablets are available for the control of vaginal candidiasis. Oral tablets and troches are used in the treatment of gastrointestinal and oral candidiasis. Combinations of nystatin with tetracycline can be used to prevent monilial overgrowth caused by the destruction of bacterial microflora of the intestine during tetracycline therapy.
Potency: ≥3000 units/mg (anhydrous).
Nystatin is stereoisomeric 33-[(3-amino-3,6-dideoxy-β-D-mannopyranosyl)
oxy]-1,3,4,7,9,11,17,37-octahydroxy-15,16,18-trimethyl-13-oxo-14,39-dioxabicyclo
[33.3.1] nonatriaconta-19,21,25,27,29,31-hexaen-36-carboxylic acid (35.1.2). Nystatin was isolated in 1949 from the products of the vital activity of the actinomycete
Streptomyces noursei, and it was the first antifungal antibiotic to be discovered. This poly�ene antibiotic is structurally similar to amphotericin B. It has a broad spectrum of activity.
The mechanism of antifungal activity is similar to the mechanism of action of ampho�tericin B. It is used for preventing and treating candida infections of the skin and mucous
membranes. In terms of preventative action, it is used for preventing the development of
candidomycosis during prolonged treatment with penicillin drugs and antibiotics of other
group, especially during oral use of tetracycline antibiotics, levomecytin, and others.
Synonyms of this drug are biofanal, moronal, nicporil, fazigin, candex, and others.
Nystatin, the first clinically useful polyene antifungal antibiotic, is a conjugated tetraene isolated from cultures of the bacterium
Streptomyces noursei in 1951. Nystatin is an effective topical antifungal against a wide variety of organisms and is available in
a variety of creams and ointments. Nystatin is too toxic to be used systemically, but because very little drug is absorbed
following oral administration, it may be administered by mouth to treat fungal infections of the mouth and gastrointestinal tract.
Poison by
intraperitoneal and intravenous routes.
Moderately toxic by subcutaneous route.
Mildly toxic by ingestion. An experimental
teratogen. Mutation data reported. An
antibiotic. When heated to decomposition it
emits toxic fumes of NOx.
Veterinary Drugs and Treatments
Orally administered nystatin is used primarily for the treatment of
oral or gastrointestinal tract Candida infections in dogs, cats, and
birds; it has been used less commonly in other species for the same
indications.
No significant gastrointestinal absorption.
Nystatin is a light yellow powder with the following solubilities at ~28o: MeOH (1.1%), ethylene glycol (0.9%), H2O (0.4%), CCl4 (0.12%), EtOH (0.12%), CHCl3 (0.05%) and *C6H6 (0.03%). It has been precipitated from MeOH solution by addition of H2O. Aqueous suspensions of this macrolide antifungal antibiotic are stable at 100o/10minutes at pH 7.0 but decompose rapidly at pH <2 and >9, and in the presence of light and O2. [Birch et al. Tetrahedron Lett 1491, 1485 1964, Weiss et al. Antibiot Chemother 7 374 1957.] It may contain a mixture of components A1, A2 and A3. [Beilstein 18 III/IV 7480.]