| Melting point |
209-217° |
| alpha |
D20 +55.9° (c = 0.27 in DMF) |
| Boiling point |
721.0±70.0 °C(Predicted) |
| Density |
1.98±0.1 g/cm3(Predicted) |
| storage temp. |
2-8°C(protect from light) |
| solubility |
DMSO (Slightly), Methanol (Slightly, Heated), Water (Slightly, Sonicated) |
| form |
Solid |
| pka |
13.07±0.70(Predicted) |
| color |
White |
| Water Solubility |
H2O: 5mg/mL, clear (warmed) |
| InChI |
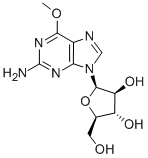
InChI=1/C11H15N5O5/c1-20-9-5-8(14-11(12)15-9)16(3-13-5)10-7(19)6(18)4(2-17)21-10/h3-4,6-7,10,17-19H,2H2,1H3,(H2,12,14,15)/t4-,6-,7+,10-/s3 |
| InChIKey |
IXOXBSCIXZEQEQ-GCTRTDOSNA-N |
| SMILES |
N1(C=NC2C(OC)=NC(N)=NC1=2)[C@H]1[C@@H](O)[C@H](O)[C@@H](CO)O1 |&1:12,13,15,17,r| |
| CAS DataBase Reference |
121032-29-9(CAS DataBase Reference) |