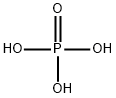
りん酸
化学名:りん酸
CAS番号.7664-38-2
英語名:Phosphoric acid
CBNumberCB3854273
MFH3O4P
MW98
MOL File7664-38-2.mol
别名
りん酸 [一般有機合成用]
オルトりん酸
りん酸水素
more
りん酸物理性質
| 融点 | ~40 °C(lit.) |
| 沸点 | 158 °C(lit.) |
| 比重(密度) | 1.685 g/mL at 25 °C(lit.) |
| 蒸気密度 | 3.4 (vs air) |
| 蒸気圧 | 2.2 mm Hg ( 20 °C) |
| 屈折率 | n |
| FEMA | 2900 | PHOSPHORIC ACID |
| 貯蔵温度 | no restrictions. |
| 溶解性 | H2O: 可溶 |
| 酸解離定数(Pka) | 2.1-7.2-12.3(at 25℃) |
| 外見 | 固体または粘性液体 |
| 比重 | 1.7 |
| 色 | ≤10(APHA) |
| PH | 3.06(1 mM solution);2.26(10 mM solution);1.63(100 mM solution); |
| 臭い (Odor) | 無臭 |
| 酸塩基指示薬変色域(pH) | 1.5 |
| 由来生物 | synthetic |
| 水溶解度 | 混和性 |
show more
| 主な危険性 | C,Xn,T,F |
| Rフレーズ | 34-35-22-39/23/24/25-36/38-23/24/25-11 |
| Sフレーズ | 7-16-26-36/37-45-36/37/39-1/2-24/25 |
| OEB | C |
| OEL | TWA: 1 mg/m3, STEL: 3 mg/m3 |
| RIDADR | UN 1805 8/PG III (PHOSPHORIC ACID, SOLUTION) |
| RIDADR | UN 3453 8/PG III (PHOSPHORIC ACID, SOLID) |
| WGK Germany | 3 |
| RTECS 番号 | TB6300000 |
| F | 3-10 |
| TSCA | TSCA listed |
| HSコード | 2809 20 00 |
| 国連危険物分類 | 8 |
| 容器等級 | III |
| ストレージクラス | 8A - Combustible corrosive hazardous materials |
| Hazard Classifications | Acute Tox. 4 Oral Eye Dam. 1 Met. Corr. 1 Skin Corr. 1B |
| 有毒物質データの | 7664-38-2(Hazardous Substances Data) |
| 毒性 | ADI 0 to 70 mg / kg (total phosphate content in terms of phosphorus, FAO / WHO, 2001). GRAS (FDA, § 182.1073, 2000). LD501530mg / kg (rat, oral). In case of daily intake of 2 ~ 4 g, it can cause mild diarrhea. The amount of sour agent used as a cola drink is 0.02% to 0.06%. |
| IDLA | 1,000 mg/m3 |
| 化審法 | (1)-422 届出不要化学物質 |
| 安衛法 | 57,57-2 |