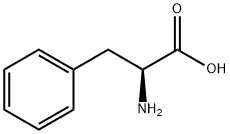
ChemicalBook > CAS DataBase List > L-Phenylalanine
L-Phenylalanine
L-Phenylalanine
- CAS No.63-91-2
- Chemical Name:L-Phenylalanine
- CBNumber:CB2488691
- Molecular Formula:C9H11NO2
- Formula Weight:165.19
- MOL File:63-91-2.mol
L-Phenylalanine Property
- Melting point 270-275 °C (dec.)(lit.)
- alpha -34.1 º (c=2, water, dry basis)
- Boiling point 293.03°C (rough estimate)
- Density 1.29
- bulk density 580kg/m3
- vapor pressure <1 Pa (25 °C)
- FEMA 3585 | L-PHENYLALANINE
- refractive index -34 ° (C=2, H2O)
- storage temp. Store below +30°C.
- solubility H2O: 0.1 M at 20 °C, clear, colorless
- form powder
- pka 2.2(at 25℃)
- color White to off-white
- PH 5.0-7.0 (25℃, 0.1M in H2O)
- Odor at 100.00 %. odorless
- Odor Type odorless
- biological source synthetic
- optical activity [α]25/D -34.2°, c = 2 in H2O (dried basis)
- Water Solubility 1-5 g/100 mL at 25 ºC
- JECFA Number 1428
- Merck 14,7271
- BRN 1910408
- Stability Stable. Incompatible with strong oxidizing agents.
- InChIKey COLNVLDHVKWLRT-QMMMGPOBSA-N
- LogP 0.24
- Substances Added to Food (formerly EAFUS) L-PHENYLALANINE
- FDA 21 CFR 172.320; 310.545
- CAS DataBase Reference 63-91-2(CAS DataBase Reference)
- EWG's Food Scores 1
- FDA UNII 47E5O17Y3R
- NIST Chemistry Reference L-Phenylalanine(63-91-2)
- EPA Substance Registry System L-Phenylalanine (63-91-2)
- Absorption cut-off at 274nm in H2O at 0.1M
- UNSPSC Code 41116107
- NACRES NA.26
Safety
- Hazard Codes :C
- Risk Statements :36/37/38-34
- Safety Statements :22-24/25-37/39-45-36/37/39-27-26
- WGK Germany :3
- RTECS :AY7535000
- F :10
- TSCA :Yes
- HS Code :29224995
- Hazardous Substances Data :63-91-2(Hazardous Substances Data)
-
NFPA 704:
0 1 0
-
Symbol(GHS)

- Signal wordWarning
- Hazard statements H315-H319-H335
- Precautionary statements P261-P305+P351+P338
L-Phenylalanine Price
More Price(38)
- Brand: Sigma-Aldrich(India)
- Product number: W358512
- Product name : L-Phenylalanine
- Purity: 99%, natural, FCC, FG
- Packaging: 1SAMPLE-K
- Price: ₹5196
- Updated: 2022/06/14
- Buy: Buy
- Brand: Sigma-Aldrich(India)
- Product number: W358512
- Product name : L-Phenylalanine
- Purity: 99%, natural, FCC, FG
- Packaging: 1KG
- Price: ₹23349.53
- Updated: 2022/06/14
- Buy: Buy
- Brand: Sigma-Aldrich(India)
- Product number: W358512
- Product name : L-Phenylalanine
- Purity: 99%, natural, FCC, FG
- Packaging: 5KG
- Price: ₹59905.55
- Updated: 2022/06/14
- Buy: Buy
- Brand: Sigma-Aldrich(India)
- Product number: W358509
- Product name : L-Phenylalanine
- Purity: 99%, FCC
- Packaging: 1SAMPLE-K
- Price: ₹4990.33
- Updated: 2022/06/14
- Buy: Buy
- Brand: Sigma-Aldrich(India)
- Product number: W358509
- Product name : L-Phenylalanine
- Purity: 99%, FCC
- Packaging: 1KG
- Price: ₹11247.18
- Updated: 2022/06/14
- Buy: Buy
L-Phenylalanine Chemical Properties,Usage,Production
- Description Phenylalanie is an essential amino acid and it is the precursor of the amino acid tyrosine. The body cannot make phenylalanie but it needs phenylalanie to produce proteins. Thus, human needs to obtain phenylalanie from food. 3 forms of phenylalanie are found in the nature: D-phenylalanine, L-phenylalanine, and DL-phenylalanine. Among these three forms, L-phenylalanine is the natural form found in most foods that containing proteins, including beef, poultry, pork, fish, milk, yogurt, eggs, cheeses, soy products, and certain nuts and seeds. The form of phenylalanine determines its application. It is suggested that L-phenylalanine can strengthen the effect of UVA radiation for people with vitiligo, in which L-phenylalanine may lead to darkening or repigmentation of the white patches, particularly on the face. The neurotransmitters L-phenylalanine affects help reduce hunger, improve memory, lessen the symptoms of ADHD and Parkinson's disease, and ease chronic pain, according to the UMMC research. Thus, L-phenylalanine is used in the manufacture of food and drink products and sold as a nutritional supplement for its reputed analgesic and antidepressant effects. L-phenylalanine is also suggested as an intermediate for anti-cancer drugs. A few small studies showed promise using L-Phenylalanine to manage alcohol withdrawal and ease PMS symptoms. It is also used in artificial sweeteners such as aspartame.
-
References
[1] http://www.umm.edu/health/medical/altmed/supplement/phenylalanine
[2] Xueqin Song , Philip L. Lorenzi , Christopher P. Landowski , Balvinder S. Vig , John M. Hilfinger , Gordon L. Amidon (2005) Amino Acid Ester Prodrugs of the Anticancer Agent Gemcitabine:? Synthesis, Bioconversion, Metabolic Bioevasion, and hPEPT1-Mediated Transport, 2, 157-167. - Chemical Properties White crystalline powder
- Chemical Properties L-Phenylalanine has no odor and a slight bitter taste. It melts with decomposition at about 283°C. The pH of a 1 in 100 solution is between 5.4 and 6.0. FEMA notes this chemical is used in cocoa substitute only.
- Occurrence Reported found in white bread, macaroni, egg noodles, corn flakes, corn grits, oatmeal, wheat bran, wheat flakes, shredded wheat, barley, brown rice, rye flour, whole grain wheat flour, buttermilk, blue cheese, cheddar cheese, cottage cheese, cream cheese, parmesan cheese, bacon, cured ham, frankfurters, pork sausage, canned red kidney beans, canned sweet corn, canned peas, canned lima beans, canned potatoes, canned asparagus, canned snap beans, canned beets, beef, lamb, fresh ham, veal round, beef liver, chicken, chicken liver, turkey and other natural sources.
- Uses phenylalanine is a conditioning agent with greater application in hair care than in skin care preparations. It is also used in suntan products.
- Uses L-Phenylalanine is an essential amino acid. L-Phenylalanine is biologically converted into L-tyrosine, another one of the DNA-encoded amino acids, which in turn is converted to L-DOPA and further conv erted into dopamine, norepinephrine, and epinephrine. L-Phenylalanine is produced for medical, feed, and nutritional applications such as in the preparation of Aspartame.
- Uses L-phenylalanine is an amino acid used as a skin-conditioning agent. It has greater use in hair care than in skin care products.
- Preparation From PTS-negative Escherichia coli bioengineered strains.
- Definition ChEBI: The L-enantiomer of phenylalanine.
-
Production Methods
In previous large-scale production processes for L-phenylalanine two enzymatic methods were applied: 1. Resolution of N-acetyl-D,L-phenylalanine by carrier-fixed microbial acylase: This process provided pharmaceutical-grade L-phenylalanine, but suffered from the disadvantage that the D-enantiomer had to be racemized and recycled. 2. Stereoselective and enantioselective addition of ammonia to trans-cinnamic acid, catalyzed by L-phenylalanine ammonia lyase (PAL, EC 4.3.1.5): PAL-containing Rhodotorula rubra was used in an industrial process to supply L-phenylalanine for the first production campaign of the sweetener aspartame. When continuously operated in an immobilized whole cell reactor, the bioconversion reached concentration up to 50 g/L Lphenylalanine at a conversion of about 83%. Other processes started from phenylpyruvate with L-aspartic acid as amine donor using immobilized cells of Escherichia coli or from a-acetamidocinnamic acid and immobilized cells of a Corynebacterium equi strain. In both cases L-phenylalanine concentrations up to 30 g/L and more (molar yields as high as at least 98 %) were reached.
However, fermentation processes based on glucose-consuming L-phenylalanine overproducing ;mutants of E. coli and coryneform strains turned out to be more economical. L-Phenylalanine is formed in ten enzymatic steps starting from erythrose-4-phosphate and phosphoenolpyruvate. A suitable profile of the specific glucose feed rate prevents acetate formation and leads to improved L-phenylalanine production with a final concentration up to 46 g/L and a corresponding yield of 18 %.
L-Phenylalanine is recovered from the fermentation broth either by two-step crystallization or by an ionexchange resin process. The preferred cell separation technique is ultrafiltration; and the filtrates may be treated with activated carbon for further purification. Instead of ion-exchange resins nonpolar, highly porous synthetic adsorbents are recommended to remove impurities. An alternative process in which a cell separator is integrated in the fermentation part, thus allowing cell recycling, was suggested for L-phenylalanine production and may lead to prospective developments.
-
Synthesis Reference(s)
Canadian Journal of Chemistry, 29, p. 427, 1951 DOI: 10.1139/v51-051
The Journal of Organic Chemistry, 30, p. 3414, 1965 DOI: 10.1021/jo01021a035
Tetrahedron Letters, 26, p. 2449, 1985 DOI: 10.1016/S0040-4039(00)94850-0 - General Description Odorless white crystalline powder. Slightly bitter taste. pH (1% aqueous solution) 5.4 to 6.
- Air & Water Reactions Water soluble. Aqueous solutions are weak acids.
- Reactivity Profile L-Phenylalanine may be light sensitive. Act as weak acids in solution.
- Health Hazard ACUTE/CHRONIC HAZARDS: When heated to decomposition L-Phenylalanine emits toxic fumes of nitrogen oxides.
- Fire Hazard Flash point data for L-Phenylalanine are not available; however, L-Phenylalanine is probably combustible.
- Biochem/physiol Actions L-Phenylalanine is an essential amino acid. It is significantly involved in the synthesis of neurotransmitters such as dopamine, epinephrine, norepinephrine, l-DOPA (Dihydroxyphenylalanine), melanin and thyroxine. L-Phenylalanine metabolism also results in phenylethylamine, that brings about effect of a stimulant in the brain and enhances mood.
- Safety Profile Mildly toxic by intraperitoneal route. An experimental teratogen. Experimental reproductive effects. Human mutation data reported. When heated to decomposition it emits toxic fumes of NOx.
- Purification Methods Likely impurities are leucine, valine, methionine and tyrosine. Crystallise L-phenylalanine from water by adding 4volumes of EtOH. Dry it in vacuo over P2O5. Also crystallise it from saturated refluxing aqueous solutions at neutral pH, or 1:1 (v/v) EtOH/water solution, or conc HCl. It sublimes at 176-184o/0.3mm with 98.7% recovery and unracemised [Gross & Gradsky J Am Chem Soc 77 1678 1955]. [Greenstein & Winitz The Chemistry of the Amino Acids J. Wiley, Vol 3 pp 2156-2175 1961, Beilstein 14 IV 1552.]
L-Phenylalanine Preparation Products And Raw materials
Raw materials
Preparation Products
Global(1028)Suppliers
- Americas 1 Australia 2 Belgium 2 Brazil 3 Canada 11 China 720 Czech 1 Czech Republic 2 Europe 5 France 2 Germany 21 Hungary 2 India 86 Italy 2 Japan 8 Mexico 1 Poland 3 Singapore 1 Slovakia 1 Slovenia 1 South Korea 3 Spain 4 Sweden 1 Switzerland 9 Thailand 1 The Netherlands 1 Tunisia 1 Ukraine 2 United Arab Emirates 1 United Kingdom 21 United States 108 USA 1 Global 1028
-
Supplier:
Hebei Longbang Technology Co., LTD
- Tel:+86-18633929156<br/>+86-18633929156
- Email:admin@hblongbang.com
- Country:China
- ProdList:972
- Advantage:58
-
Supplier:
Hebei Chuanghai Biotechnology Co., Ltd
- Tel: +8615531157085
- Email:abby@chuanghaibio.com
- Country:China
- ProdList:8808
- Advantage:58
-
Supplier:
Hebei Mujin Biotechnology Co.,Ltd
- Tel:+86 13288715578<br/>+8613288715578
- Email:sales@hbmojin.com
- Country:China
- ProdList:12809
- Advantage:58
-
Supplier:
Hebei Chuanghai Biotechnology Co,.LTD
- Tel: +86-13131129325
- Email:sales1@chuanghaibio.com
- Country:China
- ProdList:5868
- Advantage:58
-
Supplier:
Hebei Kingfiner Technology Development Co.Ltd
- Tel:+86-15532196582<br/>+86-15373005021
- Email:lisa@kingfinertech.com
- Country:China
- ProdList:3005
- Advantage:58
-
Supplier:
Guangzhou Tengyue Chemical Co., Ltd.
- Tel:+86-86-18148706580<br/>+8618826483838
- Email:evan@tyvovo.com
- Country:China
- ProdList:146
- Advantage:58
-
Supplier:
Shaanxi Haibo Biotechnology Co., Ltd
- Tel: +undefined18602966907
- Email:qinhe02@xaltbio.com
- Country:China
- ProdList:997
- Advantage:58
-
Supplier:
Anhui Ruihan Technology Co., Ltd
- Tel: +8617756083858
- Email:daisy@anhuiruihan.com
- Country:China
- ProdList:973
- Advantage:58
-
Supplier:
Henan Fengda Chemical Co., Ltd
- Tel:+86-371-86557731<br/>+86-13613820652
- Email:info@fdachem.com
- Country:China
- ProdList:20235
- Advantage:58
-
Supplier:
Hebei Saisier Technology Co., LTD
- Tel:+86-18400010335<br/>+86-18034520335
- Email:admin@hbsaisier.cn
- Country:China
- ProdList:1015
- Advantage:58
63-91-2, L-PhenylalanineRelated Search:
- Glycine 4-Nitro-3-phenyl-L-alanine D-Phenylalanine PHENYL VALERATE Phenprobamate Methyl L-phenylalaninate hydrochloride ALTRENOGEST L-Alanine Ethyl L-phenylalaninate hydrochloride L-Phenylglycinol Tris(hydroxymethyl)aminomethane L-Serine L-Threonine L-Methionine Fmoc-L-glutamic acid FMOC-LYS-OH Di-tert-butyl dicarbonate D-Tyrosine
- 63-91-2
- bc0001
- amino
- Food and Feed Additive
- Amino Acids and Derivatives
- Phenylalanine [Phe, F]
- API intermediates
- Biochemistry
- Amino Acids
- alpha-Amino Acids
- Amino Acids 13C, 2H, 15N
- Amino ACIDS SERIES
- L-Amino Acids
- Nutritional Supplements
- Amino Acids & Derivatives
- Amino Acids
- 糖醇系列/天然/人工合成甜味剂
- 中药标准品,对照品
- 抑制剂
- 营养剂
- 大化工产品
- 维生素-氨基酸
- 生物医药-医药原料
- 有机产品
- 有机化工产品
- 原料
- 临床检测标准物质
- 氨基酸系列原料
- 精细化工品
- 化学试剂
- 其他原料
- 杂质对照品
- 试剂盒-Elisa试剂盒
- 通用生化试剂-氨基酸
- 营养强化剂
- 食品添加类
- 有机化工原料
- 化工原料
- 添加剂
- 医药、农药及染料中间体
- 标准品,对照品
- 标准品 -中药标准品
- 生化试剂-氨基酸
- 对照品-中药对照品
- 医用原料
- 化工原料-1
- 化工
- 原料药
- 营养增补剂
- 食品饲料添加剂
- 生化试剂-氨基酸类
- 食品添加
- 营养强化剂-氨基酸
- 生物化工类
- 化学试剂
- 营养强化剂
- 对照品-杂质对照品
- 有机原料