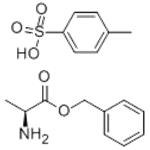
Synthesis
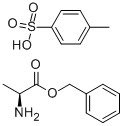
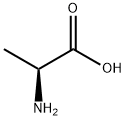
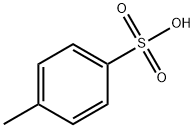
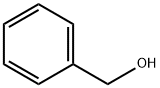
To a 300 mL three-necked flask equipped with a thermometer, stirring device, and Dean-Stark separator were added 5.00 g (56.12 mmol) L-alanine, 41.03 g (379.42 mmol) benzyl alcohol, and 12.81 g (67.34 mmol) p-toluenesulfonic acid. The system pressure was reduced from atmospheric pressure (101.3 kPa) to 2.0 kPa. The reaction mixture was heated to an internal temperature of about 50 °C to start azeotropic removal of water and slowly warmed up to 62 °C over a period of 2 hours. Maintaining the same pressure and temperature, the reaction was continued with stirring for 3.5 hours. The reaction solution was cooled to 50 °C and 111.66 g of tert-butyl methyl ether was added dropwise over 30 minutes and about 0.005 g (0.01 mmol) of L-alanine benzyl ester p-toluenesulfonate was added as a crystal seed. After keeping stirring at 50°C for 30 minutes, 55.83 g of tert-butyl methyl ether was continued dropwise over 1 hour. Stirring was continued at 50 °C for 1 h, followed by slow cooling to 0 °C over 5 h and stirring at 0 °C overnight. The crystals were collected by filtration and washed with 15.00 g of pre-cooled tert-butyl methyl ether in three portions. Drying gave 18.26 g of L-alanine benzyl ester p-toluenesulfonate in 98.6% yield based on L-alanine. The optical purity of the product was ≥99.9% e.e.
References
[1] Organic and Biomolecular Chemistry, 2012, vol. 10, # 2, p. 339 - 345
[2] Patent: EP2062873, 2009, A1. Location in patent: Page/Page column 6
[3] Amino Acids, 2017, vol. 49, # 5, p. 965 - 974
[4] Patent: US2016/97052, 2016, A1. Location in patent: Paragraph 0308
[5] Journal of Antibiotics, 1998, vol. 51, # 8, p. 786 - 794