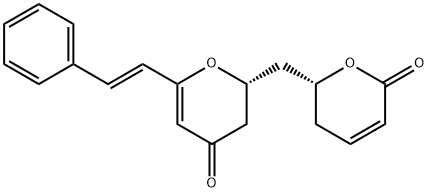
Obolactone
- Product NameObolactone
- CAS712272-88-3
- CBNumberCB92725441
- MFC19H18O4
- MW310.34
- MDL NumberMFCD32644959
- MOL File712272-88-3.mol
Chemical Properties
| Melting point | 116-118℃ |
| Boiling point | 522.2±49.0 °C(Predicted) |
| Density | 1.270±0.06 g/cm3(Predicted) |
Obolactone Price
| Product number | Packaging | Price | Product description | Buy |
|---|---|---|---|---|
| Crysdot CD32000554 | 5mg | $922 | Obolactone 95+% |
Buy |
Obolactone Chemical Properties,Usage,Production
Definition
ChEBI: Obolactone is a pyranone isolated from the trunk barks of Cryptocarya obovata and has been shown to exhibit cytotoxicity against the KB cell line. It has a role as an antineoplastic agent and a plant metabolite. It is a member of 2-pyranones and a member of 4-pyranones.Synthesis
The synthesis of (+)-obolactone (14, Scheme 3) by Brückner and Walleser employed the same conditions from the synthesis of 17-deoxyroflamycoin to transform bis-epoxide R,R-4 to bis-homoallylic diol S,S-8. Diol 8 was then protected using 2,2-dimethoxypropane under acidic conditions to provide acetonide 12 in 95% yield. One of the alkene functional groups of the C2- symmetric acetal underwent a subsequent symmetry-breaking Wacker oxidation. Treatment of acetonide 12 with catalytic PdCl2 under an atmosphere of oxygen using CuCl as the stoichiometric oxidant afforded a 64% yield of methyl ketone 13, with over-oxidation to the diketone also observed (18% yield). The methyl ketone functionality of 13 was critical for the installation of the dihydro-g-pyranone moiety in the natural product, while the syn-orientation of the C–O bonds was achieved through Mitsunobu inversion of the lactone stereocenter. Brückner and Walleser specifically mention that while Krische and co-workers have reported on an impressive single-step procedure for the catalytic enantioselective synthesis of bishomoallylic diol (S,S-4) from 1,3-propanediol, and have used this method extensively in the synthesis of polyketide natural products, 21,22 the high cost of catalyst and ligand precluded their use on scale in this case.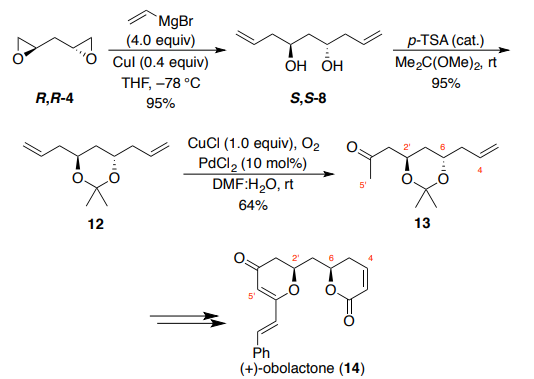
Preparation Products And Raw materials
Obolactone Suppliers
Global(14)Suppliers
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| support@targetmol.com | United States | 38630 | 58 | ||
| 0871-65217109 13211707573; |
y.liu@mail.biobiopha.com | China | 5653 | 65 | |
| 028-81700200 18116577057 |
3003855609@qq.com | China | 7787 | 56 | |
| 0510-85629785 18013409632 |
sales@reading-chemicals.com | China | 15178 | 58 | |
| 888-539-0666 | info@emmx.com | United States | 8447 | 60 | |
| 021-61312847; 18021002903 |
3008007409@qq.com | China | 27313 | 60 | |
| 18301908279 18301908279 |
1292752157@qq.com | China | 7744 | 58 | |
| 021-57763112 13585886131 |
3004987436@qq.com | China | 9693 | 58 | |
| 0510-85629785 18013409632 |
sales@reading-chemicals.com | China | 14081 | 58 | |
| 400-8087372 13161114455 |
puxi_sale@qq.com | China | 4990 | 58 |
712272-88-3, ObolactoneRelated Search
PROMPT×
PROMPT
The What'sApp is temporarily not supported in mainland China
The What'sApp is temporarily not supported in mainland China
Cancel
Determine