Synthesis
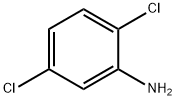
General procedure for the synthesis of 2,5-dichlorofluorobenzene from 2,5-dichloroaniline:
1. Continuous diazotization step: material A (50 mL aqueous solution containing 2,5-dichloroaniline (100 mmol), fluoboric acid (120 mmol) and hydrochloric acid (180 mmol)) was pumped through a T-fitting into a reaction tube at a flow rate of 4 mL/min at 25°C with material B (50 mL aqueous solution containing sodium nitrite (105 mmol)), the mixture flowed through the outlet and collected in a cooling vessel. Vigorous stirring was maintained. The slurry was cooled to -5 °C and then diafiltrated. The solid was washed with methanol and subsequently dried under vacuum to give 2,5-dichlorobenzenediazonium tetrafluoroborate.
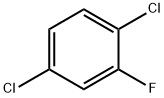
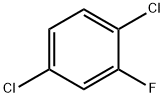
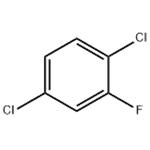
2. Continuous fluorination step: a slurry of 2,5-dichlorobenzenediazonium tetrafluoroborate prepared above in 300 mL of co-solvent was introduced continuously into a reaction tube at a flow rate of 4 mL/min. The mixture was kept at the set temperature for 1 min and subsequently cooled in the tandem tube. The collected liquid was washed with aqueous NaOH and water to give an almost colorless liquid of 2,5-dichlorofluorobenzene.
References
[1] Tetrahedron Letters, 2013, vol. 54, # 10, p. 1261 - 1263
[2] Journal of the American Chemical Society, 1959, vol. 81, p. 94,95, 97
[3] Journal of the American Chemical Society, 1953, vol. 75, p. 4590
[4] Acta Chimica Academiae Scientiarum Hungaricae, 1957, vol. 10, p. 227,229