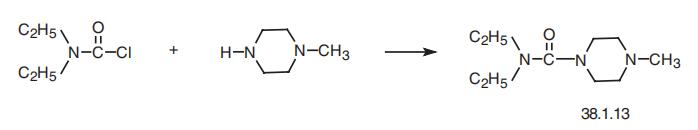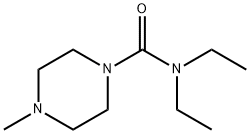
N,N-Diethyl-4-methyl-1-piperazincarboxamid
Bezeichnung:N,N-Diethyl-4-methyl-1-piperazincarboxamid
CAS-Nr90-89-1
Englisch Name:DIETHYLCARBAMAZINE
CBNumberCB9243620
SummenformelC10H21N3O
Molgewicht199.29
MOL-Datei90-89-1.mol
Synonyma
Diethylcarbamazin
N,N-Diethyl-4-methyl-1-piperazincarboxamid
N,N-Diethyl-4-methyl-1-piperazincarboxamid physikalisch-chemischer Eigenschaften
| Schmelzpunkt | 65-67℃ |
| Siedepunkt | 127-129℃ (10 Torr) |
| Dichte | 1.0173 (rough estimate) |
| Brechungsindex | 1.4930 (estimate) |
| storage temp. | Sealed in dry,Room Temperature |
| pka | pKa 7.7 (Uncertain) |
| EPA chemische Informationen | Diethylcarbamazine (90-89-1) |