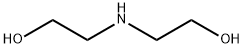
Diethanolamin
Bezeichnung:Diethanolamin
CAS-Nr111-42-2
Englisch Name:Diethanolamine
CBNumberCB5852839
SummenformelC4H11NO2
Molgewicht105.14
MOL-Datei111-42-2.mol
Synonyma
2,2'-Iminodiethanol
Diethanolamin
DEA
2,2'-Dihydroxydiethylamin
Diethanolamin physikalisch-chemischer Eigenschaften
| Schmelzpunkt | 28 °C (lit.) |
| Siedepunkt | 217 °C/150 mmHg (lit.) |
| Dichte | 1.097 g/mL at 25 °C (lit.) |
| Dampfdichte | 3.6 (vs air) |
| Dampfdruck | <0.98 atm ( 100 °C) |
| Brechungsindex | n |
| Flammpunkt | 280 °F |
| storage temp. | 2-8°C |
| Löslichkeit | H2O: 1 M at 20 °C, clear, colorless |
| Aggregatzustand | Viscous Liquid or Low Melting Solid |
| Wichte | 1.09 |
| Farbe | APHA: ≤15 |
| PH | 11.0-12.0 (25℃, 1M in H2O) |
| pka | 8.88(at 25℃) |
| Geruch (Odor) | Mild ammoniacal; faint, fishy; characteristic. |
| Explosionsgrenze | 2.1-10.6%(V) |
| Wasserlöslichkeit | MISCIBLE |
| Sensitive | Hygroscopic |
| maximale Wellenlänge (λmax) | λ: 260 nm Amax: 0.04 λ: 280 nm Amax: 0.02 |
| Merck | 14,3107 |
| BRN | 605315 |
| Expositionsgrenzwerte | TLV-TWA 3 ppm (~13 mg/m3) (ACGIH). |
| Dielectric constant | 2.8(25℃) |
| Stabilität | Stable. Incompatible with carbon dioxide, strong acids, strong oxidizing agents. Deliquescent. |
| InChIKey | ZBCBWPMODOFKDW-UHFFFAOYSA-N |
| LogP | -2.46 at 25℃ |
| Oberflächenspannung | 47.2mN/m at 293.15K |
| CAS Datenbank | 111-42-2(CAS DataBase Reference) |
| IARC | 2B (Vol. 77, 101) 2013 |
| NIST chemische Informationen | Diethanolamine(111-42-2) |
| EPA chemische Informationen | Diethanolamine (111-42-2) |
| Kennzeichnung gefährlicher | Xn |
| R-Sätze: | 22-38-41-48/22 |
| S-Sätze: | 26-36/37/39-46 |
| RIDADR | 3267 |
| OEB | A |
| OEL | TWA: 3 ppm (15 mg/m3) |
| WGK Germany | 1 |
| RTECS-Nr. | KL2975000 |
| F | 3 |
| Selbstentzündungstemperatur | 689 °F |
| TSCA | Yes |
| PackingGroup | II |
| HS Code | 29221200 |
| Giftige Stoffe Daten | 111-42-2(Hazardous Substances Data) |
| Toxizität | LD50 orally in rats: 12.76 g/kg (Smyth) |
Gefahreninformationscode (GHS)
-
Bildanzeige (GHS)



-
Alarmwort
Achtung
-
Gefahrenhinweise
H302:Gesundheitsschädlich bei Verschlucken.
H315:Verursacht Hautreizungen.
H318:Verursacht schwere Augenschäden.
H373:Kann die Organe schädigen bei längerer oder wiederholter Exposition.
-
Sicherheit
P202:Vor Gebrauch alle Sicherheitshinweise lesen und verstehen.
P280:Schutzhandschuhe/Schutzkleidung/Augenschutz tragen.
P301+P312:BEI VERSCHLUCKEN: Bei Unwohlsein GIFTINFORMATIONSZENTRUM/Arzt/... (geeignete Stelle für medizinische Notfallversorgung vom Hersteller/Lieferanten anzugeben) anrufen.
P302+P352:BEI BERÜHRUNG MIT DER HAUT: Mit viel Wasser/... (Hersteller kann, falls zweckmäßig, ein Reinigungsmittel angeben oder, wenn Wasser eindeutig ungeeignet ist, ein alternatives Mittel empfehlen) waschen.
P305+P351+P338:BEI KONTAKT MIT DEN AUGEN: Einige Minuten lang behutsam mit Wasser spülen. Eventuell vorhandene Kontaktlinsen nach Möglichkeit entfernen. Weiter spülen.
P308+P313:BEI Exposition oder falls betroffen: Ärztlichen Rat einholen/ärztliche Hilfe hinzuziehen.
Diethanolamine Chemische Eigenschaften,Einsatz,Produktion Methoden
-
ERSCHEINUNGSBILD
WEISSE KRISTALLEODER FARBLOSE VISKOSE HYGROSKOPISCHE FLüSSIGKEIT MIT CHARAKTERISTISCHEM GERUCH. -
PHYSIKALISCHE GEFAHREN
Die Dämpfe sind schwerer als Luft. -
CHEMISCHE GEFAHREN
Zersetzung beim Verbrennen unter Bildung giftiger Rauche. Mittelstarke Base in wässriger Lösung. Reagiert sehr heftig mit starken Oxidationsmittelnund starken Säuren. Greift Kupfer, Zink, Aluminium und ihre Legierungen an. -
ARBEITSPLATZGRENZWERTE
TLV: 2 mg/m?(als TWA); Hautresorption; (ACGIH 2005).
MAK: 1 mg/m?(Einatembare Fraktion); Spitzenbegrenzung: überschreitungsfaktor I(1); Hautresorption; Sensibilisierung der Haut; Krebserzeugend Kategorie 3B; Schwangerschaft: Gruppe C (DFG 2007).
-
AUFNAHMEWEGE
Aufnahme in den Körper durch Inhalation der Dämpfe und durch Verschlucken. -
INHALATIONSGEFAHREN
Beim Verdampfen bei 20°C tritt eine gesundheitsschädliche Kontamination der Luft nicht oder nur sehr langsam ein. -
WIRKUNGEN BEI KURZZEITEXPOSITION
WIRKUNGEN BEI KURZZEITEXPOSITION:
Die Substanz verätzt die Augen. -
WIRKUNGEN NACH WIEDERHOLTER ODER LANGZEITEXPOSITION
Wiederholter oder andauernder Kontakt kann zu Hautsensibilisierung führen. Möglich sind Auswirkungen auf Leberund Nieren. -
LECKAGE
Verschüttetes Material in abdichtbaren Behältern sammeln; falls erforderlich durch Anfeuchten Staubentwicklung verhindern. An sicheren Ort bringen. Persönliche Schutzausrüstung: Atemschutzgerät, A/P2-Filter für organische Dämpfe und schädlichen Staub. -
R-Sätze Betriebsanweisung:
R22:Gesundheitsschädlich beim Verschlucken.
R38:Reizt die Haut.
R41:Gefahr ernster Augenschäden.
R48/22:Gesundheitsschädlich: Gefahr ernster Gesundheitsschäden bei längerer Exposition durch Verschlucken. -
S-Sätze Betriebsanweisung:
S26:Bei Berührung mit den Augen sofort gründlich mit Wasser abspülen und Arzt konsultieren.
S36/37/39:Bei der Arbeit geeignete Schutzkleidung,Schutzhandschuhe und Schutzbrille/Gesichtsschutz tragen.
S46:Bei Verschlucken sofort ärztlichen Rat einholen und Verpackung oder Etikett vorzeigen. -
Aussehen Eigenschaften
C4H11NO2; ß,ß'-Dihydroxydiethylamin, Bis-(ß-hydroxylethyl)-amin. Farblose fest oder flüssige Substanz (Schmelzpunkt 28鳦) mit schwach aminartigem Geruch. -
Gefahren für Mensch und Umwelt
Reizt die Augen und die Haut. Inhalation führt zu Reizungen und Atemnot.
Nicht mit Oxidationsmitteln, Nitrilen, salpetriger Säure, Säuren und Anhydriden in Berührung bringen. -
Schutzmaßnahmen und Verhaltensregeln
Schutzhandschuhe als kurzzeitiger Spritz- Staubschutz. -
Verhalten im Gefahrfall
Mit flüssigkeitsbindendem Material, z.B Rench Rapid aufnehmen. Der Entsorgung zuführen. Nachreinigen.
Kohlendioxid, Wasser, Schaum, Pulver.
Brennbar. Im Brandfall können nitrose Gase freigesetzt werden. Mit Luft Bildung explosionsfähiger Gemische möglich. -
Erste Hilfe
Nach Hautkontakt: Mit reichlich Wasser abwaschen.
Nach Augenkontakt: Mit reichlich Wasser bei geöffnetem Lidspalt mindestens 15 Minuten ausspülen. Sofort Augenarzt hinzuziehen.
Nach Einatmen: Frischluft. Arzt hinzuziehen.
Nach Verschlucken: Reichlich Wasser trinken lassen. Erbrechen auslösen. Arzt hinzuziehen.
Nach Kleidungskontakt: Kontaminierte Kleidung sofort entfernen.
Ersthelfer: siehe gesonderten Anschlag -
Sachgerechte Entsorgung
Als halogenfreie, organische Lösemittelabfälle. -
Beschreibung
Diethanolamine is an organic base which has been used as an emulsifying and dispersing agent.It can also be used as a basic buffer, with optimal pH about pH 9, if titrated with HCl or other acid. Other uses include: to "scrub" gases, as a chemical intermediate, as humectant or softening agent. -
Chemische Eigenschaften
The USP32–NF27 describes diethanolamine as a mixture of ethanolamines consisting largely of diethanolamine. At about room temperature it is a white, deliquescent solid. Above room temperature diethanolamine is a clear, viscous liquid with a mildly ammoniacal odor.

Diethanolamine is used as surface-active agent in metal-cutting fluids and oils, as a corrosion inhibitor, as a dispersant in agricultural chemical formulations, and as an intermediate in the production of other compounds such as fatty acid condensates of diethanolamine which are extensively used in soaps and cosmetics as emulsifiers, thickeners, wetting agents and detergents (Beyer et al., 1983). In the cosmetic formulations, the concentration of diethanolamine may range from 1 to 25% (National Toxicology Program, 1999a). -
Verwenden
Diethanolamine similar to triethanolamine (T775580) is used as a surfactant. It also has the potential to be a corrosion inhibitor by means of chemisorption. -
Verwenden
To scrub gases as indicated under ethanolamine. Diethanolamine can be used with cracking gases and coal or oil gases which contain carbonyl sulfide that would react with monoethanolamine. As rubber chemicals intermediate. In the manufacture of surface active agents used in textile specialties, herbicides, petroleum demulsifiers. As emulsifier and dispersing agent in various agricultural chemicals, cosmetics, and pharmaceuticals. In the production of lubricants for the textile industry. As humectant and softening agent. In organic syntheses. -
Verwenden
Diethanolamine is used in the production ofsurface-active agents and lubricants for thetextile industry; as an intermediate for rubberchemicals; as an emulsifier; as a humectantand softening agent; as a detergent in paints,shampoos, and other cleaners; and as anintermediate in resins and plasticizers. -
synthetische
Diethanolamine is prepared commercially by the ammonolysis of ethylene oxide. The reaction yields a mixture of monoethanolamine, diethanolamine, and triethanolamine which is separated to obtain the pure products. -
Vorbereitung Methode
Diethanolamine is produced with monoethanolamine and triethanolamine by ammonolysis of ethylene oxide; diethanolamine is then separated by distillation (Mullins 1978). In 1984, 166.2 million pounds of diethanolamine were produced in the United States (USTIC 1985). -
Definition
ChEBI: A member of the class of ethanolamines that is ethanolamine having a N-hydroxyethyl substituent. -
Allgemeine Beschreibung
Oily colorless liquid or solid white crystals. Slight rotten fish or ammonia odor. Denser than water. -
Air & Water Reaktionen
Water soluble. -
Reaktivität anzeigen
2,2'-Iminodiethanol is an aminoalcohol. Amines are chemical bases. They neutralize acids to form salts plus water. These acid-base reactions are exothermic. The amount of heat that is evolved per mole of amine in a neutralization is largely independent of the strength of the amine as a base. Amines may be incompatible with isocyanates, halogenated organics, peroxides, phenols (acidic), epoxides, anhydrides, and acid halides. Flammable gaseous hydrogen is generated by amines in combination with strong reducing agents, such as hydrides. 2,2'-Iminodiethanol is hygroscopic. 2,2'-Iminodiethanol may be sensitive to exposure to air and light. 2,2'-Iminodiethanol can react with oxidizing materials, acids, CO2, copper alloys, aluminum, zinc, galvanized iron and copper. -
Health Hazard
The irritant action of diethanolamine on theeyes can be severe. Direct contact of thepure liquid can impair vision. Irritation onthe skin may be mild to moderate. Theacute oral toxicity of this compound waslow in test animals. The toxic symptomsinclude somnolence, excitement, and musclecontraction.
LD50 value, oral (mice): 3300 mg/kg
The vapor pressure of diethanolamine isnegligibly low (<0.01 torr at 20°C (68°F)).At ordinary temperature, this compoundshould not cause any inhalation hazard. Themists, fumes, or vapors at high temperatures,however, can produce eye, skin, and respiratory tract irritation.
In contrast to monoethanolamine, dieth anolamine administered to mice at 1125 mg/kg/day caused no change in maternal mortality, litter size, or percentage survival of thepups (Environmental Health Research andTesting 1987). -
Brandgefahr
Special Hazards of Combustion Products: Irritating vapors are generated when heated. -
Flammability and Explosibility
Non flammable -
Chemische Reaktivität
Reactivity with Water : No reaction; Reactivity with Common Materials: No reaction; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Flush with water; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent. -
Pharmazeutische Anwendungen
Diethanolamine is primarily used in pharmaceutical formulations as a buffering agent, such as in the preparation of emulsions with fatty acids. In cosmetics and pharmaceuticals it is used as a pH adjuster and dispersant.
Diethanolamine has also been used to form the soluble salts of active compounds, such as iodinated organic acids that are used as contrast media. As a stabilizing agent, diethanolamine prevents the discoloration of aqueous formulations containing hexamethylenetetramine-1,3-dichloropropene salts.
Diethanolamine is also used in cosmetics. -
Industrielle Verwendung
Diethanolamine undergoes reactions characteristic of secondary amines and of alcohols. Two industrially important reactions of the ethanolamines involve reaction with carbon dioxide or hydrogen sulfide to yield water soluble salts, and reaction with long chain fatty acids to form neutral ethanolamine soaps (Mullins 1978). Substituted ethanolamine compounds, such as soaps, are used extensively as emulsifiers, thickeners, wetting agents, and detergents in cosmetic formulations (including skin cleaners, creams, and lotions) (Beyer et al 1983).
Diethanolamine is used as a dispersing agent in various agricultural chemicals, as an absorbent for acidic gases (hydrogen sulfide and carbon dioxide), as a humectant, as an intermediate in the synthesis of morpholine, as a surface-active agent in cutting fluids, as a corrosion inhibitor, as a component in textile specialty agents, and as a secondary vulcanization accelerator in the rubber industry. Diethanolamine is also used in cleaners and pharmaceutical ointments, in polyurethane formulations, in herbicides, and in a variety of organic syntheses (Beyer et al 1983; Mullins 1978; Windholz 1983). Diethanolamine is permitted in articles intended for use in the production, processing, or packaging of food (CFR 1981), and is permitted as a secondary direct food additive from use in delinting cottonseed in the production of cottonseed oil or meal cake (Fed. Reg. 1982). Because of the wide industrial and consumer uses, large amounts of this chemical are discharged into water and sewage in an unaltered form (Yordy and Alexander 1981). -
Kontakt-Allergie
Diethanolamine is contained in many products, as a metalworking fuid. Traces may exist in other etha- nolamine-containing fuids. -
Sicherheitsprofil
Poison by intraperitoneal route. Moderately toxic by ingestion and subcutaneous routes. Mildly toxic by skin contact. A severe eye and mild skin irritant. Experimental reproductive effects. Combustible when exposed to heat or flame; can react with oxidizing materials. To fight fire, use alcohol foam, water, Co2, dry chemical. When heated to decomposition it emits toxic fumes such as NOx. See also AMINES. -
Sicherheit(Safety)
Diethanolamine is used in topical and parenteral pharmaceutical formulations, with up to 1.5% w/v being used in intravenous infusions. Experimental studies in dogs have shown that intravenous administration of larger doses of diethanolamine results in sedation, coma, and death.
Animal toxicity studies suggest that diethanolamine is less toxic than monoethanolamine, although in rats the oral acute and subacute toxicity is greater. Diethanolamine is said to be heptacarcinogenic in mice and has also been reported to induce hepatic choline deficiency in mice.
Diethanolamine is an irritant to the skin, eyes, and mucous membranes when used undiluted or in high concentration. However, in rabbits, aqueous solutions containing 10% w/v diethanolamine produce minor irritation. The lethal human oral dose of diethanolamine is estimated to be 5–15g/kg body-weight.
The US Cosmetic Ingredient Review Expert Panel evaluated diethanolamine and concluded that it is safe for use in cosmetic formulations designed for discontinuous, brief use followed by thorough rinsing from the surface of the skin. In products intended for prolonged contact with the skin, the concentration of ethanolamines should not exceed 5%. Diethanolamine should not be used in products containing N-nitrosating agents.
LD50 (guinea pig, oral): 2.0g/kg
LD50 (mouse, IP): 2.3g/kg
LD50 (mouse, oral): 3.3g/kg
LD50 (rabbit, skin): 12.2g/kg
LD50 (rat, IM): 1.5g/kg
LD50 (rat, IP): 0.12g/kg
LD50 (rat, IV): 0.78g/kg
LD50 (rat, oral): 0.71g/kg
LD50 (rat, SC): 2.2g/kg -
mögliche Exposition
Diethanolamine is present in machining and grinding fluids and has been detected in workplace air in the metal manufacturing industry. It was present in bulk cutting fluids at levels ranging from 4 to 5% (Kenyon et al., 1993). Diethanolamine has also been reported to be present in wetting fluids used in road paving. A level of 0.05 mg/m3 was detected in a stationary sample at a slurry machine discharging a bitumen emulsion containing 0.2% of the amine. All personal exposures were below the detection limit (0.02 mg/m3) (Levin et al., 1994). In a German study (1992–94), diethanolamine was measured in samples of metalworking fluids in a range of 0–44% (n = 69). The number of samples with diethanolamine present steadily declined from 90% to 60% over the study period (Pfeiffer et al., 1996). -
Erste Hilfe
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. Give large quantities of water and inducevomiting. Do not make an unconscious person vomit. -
Carcinogenicity
When DEA was administered cutaneously to pregnant rats and rabbits during organogenesis, developmental toxicity (skeletal variations) was observed only in the rat and only at doses causing significant maternal toxicity.
The 2003 ACGIH threshold limit valuetime- weighted average (TLV-TWA) is 3ppm (13mg/m3). -
Stoffwechsel
Treatment of Wistar or Sherman rats with diethanolamine caused increases in the formation of hepatic phospholipids (Artom et al 1949). In addition, dietary administration led to incorporation of ethanolamine into hepatic phospholipids (Artom et al 1949), and repeated oral administration of diethanolamine in drinking water (one to three wk) at a dose of 320 mg/kg/d was found to reduce the level of incorporation of ethanolamine and choline into hepatic and renal phospholipids in Sprague-Dawley rats (Barbee and H?rtung 1979b).
Dermal absorption of diethanolamine is suggested to occur in rats since Nnitrosodiethanolamine was excreted in the urine of male Sprague-Dawley rats which had been administered diethanolamine by dermal application and given nitrite in their drinking water (Preussman et al 1981). -
Lager
Diethanolamine is hygroscopic and light- and oxygen-sensitive; it should be stored in an airtight container, protected from light, in a cool, dry place. -
Versand/Shipping
UN2491 Ethanol amine or Ethanolamine solutions, Hazard class: 8; Labels: 8-Corrosive material. -
läuterung methode
Fractionally distil the amine twice, then fractionally crystallise it from its melt. Its solubility in H2O is 10% at 20o. [Perrin & Dempsey Buffers for pH and Metal Ion Control Chapman & Hall, London 1974, Beilstein 4 H 283, 4 II 729, 4 III 689, 4 IV 1514.] -
Inkompatibilitäten
Diethanolamine is a secondary amine that contains two hydroxy groups. It is capable of undergoing reactions typical of secondary amines and alcohols. The amine group usually exhibits the greater activity whenever it is possible for a reaction to take place at either the amine or a hydroxy group.
Diethanolamine will react with acids, acid anhydrides, acid chlorides, and esters to form amide derivatives, and with propylene carbonate or other cyclic carbonates to give the corresponding carbonates. As a secondary amine, diethanolamine reacts with aldehydes and ketones to yield aldimines and ketimines. Diethanolamine also reacts with copper to form complex salts. Discoloration and precipitation will take place in the presence of salts of heavy metals. -
Filter für giftige stoffe
The initial threshold screening level (ITSL) for diethanolamine (DEA) is 0.2 μg/m3 (annual averaging time) and an acute ITSL for diethanolamine is 10 μg/m3 (8-hour averaging time). -
Waste disposal
Controlled incineration; incinerator equipped with a scrubber or thermal unit to reduce nitrogen oxides emissions -
Regulatory Status
Included in the FDA Inactive Ingredients Database (IV infusions, ophthalmic solutions, and topical preparations). Included in medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Diethanolamine Upstream-Materialien And Downstream Produkte
Upstream-Materialien
Downstream Produkte
- softener 101
- coconut oil biethyl alcohol acylamine 1︰1 type
- fatliquor RCF I/II
- 4-(PIPERIDINE-1-CARBONYL)PHENYLBORONIC ACID
- Tetranatrium-4,4'-bis[[4-[bis(2-hydroxyethyl)amino]-6-(4-sulfonatoanilino)-1,3,5-triazin-2-yl]amino]stilben-2,2'-disulfonat]
- 4-(2-CYANOETHYLAMINOCARBONYL)PHENYLBORONIC ACID
- antistatic Agent P
- 3-(2-CYANOETHYLAMINOCARBONYL)PHENYLBORONIC ACID
- 1-(3-Chlorphenyl)-4-(3-chlorpropyl)piperaziniumchlorid
- PIPERAZINE HEXAHYDRATE
- MORPHOLINEFATTYACIDSALT
- 1-(3-Chlorphenyl)piperazindihydrochlorid
- 2,2'-Methyliminodiethanol
- coconut oil alcohol acylamide
- Ethyl-4-phenylpiperidin-4-carboxylat
- N-(2-[4-(4-CHLOROPHENYL)PIPERAZIN-1-YL]ETHYL)-3-METHOXYBENZAMIDE
- 2-Methoxy-ethanol
- 2-[[4-[[4-[Bis(2-hydroxyethyl)amino]-6-chlor-1,3,5-triazin-2-yl]amino]phenyl]azo]-p-kresol
- Desulfurizer,high efficiency
- Cyclen
- dodecylyl diethanol amide 1︰2 type
- Cyclophosphamide monohydrate
- 4-Phenylpiperidin-4-carbonitrilhydrochlorid
- N,N-Bis(2-chlorethyl)-p-toluolsulfonamid
- 2-Bromo-N-(2-bromoethyl)ethanamine
1of8
Diethanolamin Anbieter Lieferant Produzent Hersteller Vertrieb Händler.
Global(900)Suppliers
Region
-
Shandong Dexiang International Trade Co., Ltd
Telefon +86-15662691337<br/>+86-15662695772
E-Mail 539942812@qq.com
-
Hebei Weibang Biotechnology Co., Ltd
Telefon +8615531157085
E-Mail abby@weibangbio.com
-
Hebei Mujin Biotechnology Co.,Ltd
Telefon +86 13288715578<br/>+8613288715578
E-Mail sales@hbmojin.com
-
Hebei Chuanghai Biotechnology Co,.LTD
Telefon +86-13131129325
E-Mail sales1@chuanghaibio.com
-
Henan Fengda Chemical Co., Ltd
Telefon +86-371-86557731<br/>+86-13613820652
E-Mail info@fdachem.com
-
Qingdao RENAS Polymer Material Co., Ltd.
Telefon +86-0532-86867058<br/>+86-18562606086
E-Mail sales@qdrenas.com
-
Henan Tianfu Chemical Co.,Ltd.
Telefon +86-0371-55170693<br/>+86-19937530512
E-Mail info@tianfuchem.com
-
Hefei TNJ Chemical Industry Co.,Ltd.
Telefon +86-0551-65418679<br/>+8618949832763
E-Mail info@tnjchem.com
-
Tianjin Zhongxin Chemtech Co., Ltd.
Telefon 86-22-66880623<br/>+8618622897568
E-Mail sales@tjzxchem.com
-
Shanxi Naipu Import and Export Co.,Ltd
Telefon +86-13734021967<br/>+8613734021967
E-Mail kaia@neputrading.com
1of2
111-42-2, Diethanolamine Verwandte Suche:
- 2-Aminoethanol
- 2,2'-(3-Chlorphenylimino)diethanol
- 2,2',2'',2'''-Ethylendinitrilotetraethanol
- 2,2'-Methyliminodiethanol
- 2,2',2''-Nitrilotriethanol
- N,N'-1,2-Ethandiylbis(N-(carboxymethyl)glycin,Dinatrium-Salz
- trans-Cyclohexan-1,2-dinitrilotetraessigsure
- Hexamethylendiamintetraessigsure
- 1,1'-Iminodipropan-2-ol
- N-(2-Hydroxyethyl)ethylendiamintriessigsure
1of4
