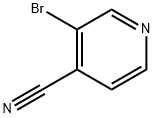
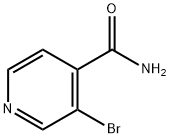
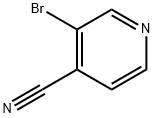
3-Bromopyridine-4-carboxamide (687 mg, 3.4 mmol) was used as a raw material and was slowly added to phosphoryl chloride (5.0 mL) at 0 °C. The reaction mixture was then stirred under reflux conditions for 2 hours. Upon completion of the reaction, the mixture was cooled to room temperature and slowly poured into ice (100 g) under stirring and neutralized with 5 M aqueous sodium hydroxide solution (60 mL). The neutralized mixture was extracted with ether (2 x 50 mL). The organic layers were combined and sequentially washed with saturated aqueous sodium bicarbonate solution, treated with activated carbon and filtered. Subsequently, the solvent was removed by evaporation to give the crude product. The crude product was purified by recrystallization from petroleum ether to afford the target compound 3-bromo-4-cyanopyridine (292 mg, 42% yield) as an off-white powder. The product characterization data were as follows: melting point 79-81 °C (literature value 96.6-98.2 °C); 1H NMR (CDCl3) δ 7.53 (1H, d, J = 4.9 Hz, 5-H), 8.69 (1H, d, J = 4.9 Hz, 6-H), 8.92 (1H, s, 2-H); 13C NMR (CDCl3) (HSQC/HMBC ) δ 114.79 (C≡N), 122.14 (4-C), 124.17 (3-C), 126.75 (5-C), 148.76 (6-C), 152.69 (2-C); MS (ESI) m/z 182.9552 (M + H) (C6H4N2 79Br) Theoretical value 182.9541.