The activity of Crizotinib in lung cancer
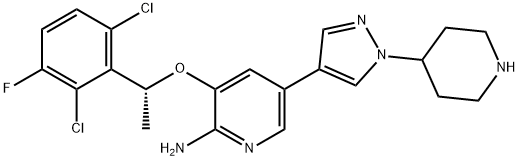
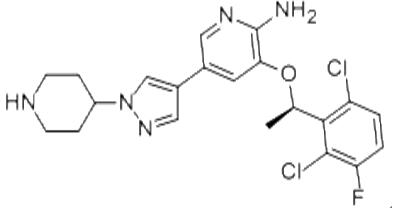
Crizotinib is described chemically as (R)-3-[l-(2,6-Dichloro-3-fluorophenyl) ethoxy]-5-[1-(piperidin-4-yl)-1H-pyrazol-4-yl] pyridin-2-amine (scheme 1), and it is an anti-cancer drug acting as an ALK (anaplastic lymphoma kinase) and ROS1 (c-ros oncogene 1) inhibitor, approved for treatment of some non-small cell lung carcinoma (NSCLC), and undergoing clinical trials testing its safety and efficacy in anaplastic large cell lymphoma, neuroblastoma, and other advanced solid tumors in both adults and children [1].
In the treatment of lung cancer, the formation of ALK fusion proteins results in the activation and dysregulation of the gene expression and signaling, which can contribute to increased cell proliferation and survival in tumors expressing these proteins. Crizotinib demonstrates concentration-dependent inhibition of ALK and c-Met phosphorylation in cell-based assays using tumor cell lines, and also demonstrates antitumor activity in mice bearing tumor xenografts. [2].
Scheme 1 Diagrammatic representation of the structure of Crizotinib
The antitumor efficacy of crizotinib was initially demonstrated in two multicenter, single-arm studies. These studies included 255 patients, all of whose tumors contained an ALK gene rearrangement, and 95% of the patients had metastatic disease and 5% had locally advanced NSCLC. Overall, 94% of the patients had received prior systemic therapy for advanced or metastatic disease, and 76% had received two or more treatment regimens. The combined objective (complete plus partial) response rate was 55%.
Evidence of the impact of crizotinib on the survival of patients came from a non-randomized, retrospective analysis of the patients enrolled in the phase I study. The one- and two-year survival rates for patients treated with crizotinib were 74 and 54%, respectively, with a median follow-up of 18 months. In a cohort of 36 patients with the ALK fusion oncogene, who were not treated with crizotinib, the survival rates at one- and two-years were 44% and 12%, respectively. A comparison with a larger cohort of patients with wild-type tumors did not identify a difference when compared with those with the ALK rearrangement not treated with crizotinib, suggesting that the presence of ALK rearrangement was not prognostically significant.
The activity of crizotinib in ALK rearrangement positive NSCLC was subsequently confirmed in a phase III trial, in which 347 patients were randomly assigned to crizotinib or single agent chemotherapy with either pemetrexed or docetaxel. Patients with progressive disease, on chemotherapy, were allowed to cross over and receive treatment with crizotinib. All patients had received one prior platinum-based regimen. Treatment with crizotinib significantly increased progression-free survival, the primary endpoint of the trial, compared to chemotherapy. There was no significant difference in the overall survival, but 64% of the chemotherapy-treated patients had crossed over to crizotinib.
Thus, crizotinib is a straightforward, biology-based biomarker, predicting a high response rate in heavily pre-treated patients and is relatively non-toxic. It is a triumph for a targeted therapy in the era of oncogenic driver mutations.
Patients with anaplastic lymphoma kinase (ALK) gene rearrangements often manifest dramatic responses to crizotinib, a small molecule ALK inhibitor. However, part of patients respond and acquired drug resistance inevitably some time, so it should be noticed also [3].
Reference
[1] https://en.wikipedia.org/wiki/Crizotinib
[2] Arvind Sahu,etc. Crizotinib: A comprehensive review, South Asian J Cancer. 2013 Apr-Jun; 2(2): 91–97.
[3] Robert C. Doebele, etc., Mechanisms of Resistance to Crizotinib in Patients with ALK Gene Rearranged Non-Small Cell Lung Cancer, Clin Cancer Res. 2012 Mar 1; 18(5): 1472–1482.
Related articles And Qustion
See also
Lastest Price from Crizotinib manufacturers

US $5.00-0.50/KG2025-06-05
- CAS:
- 877399-52-5
- Min. Order:
- 1KG
- Purity:
- 99% hplc
- Supply Ability:
- 500TONS

US $0.00/kg2025-04-29
- CAS:
- 877399-52-5
- Min. Order:
- 1kg
- Purity:
- 0.99
- Supply Ability:
- 1000kg