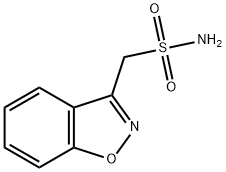
Zonisamide is a new generation of sulfonamide anticonvulsant that is primarily used as supplemental therapy in treatment of partial seizures in combination with other antiepileptic medications. Besides, it is approved to be applied as an adjunctive therapy in adults suffering from infantile spasm, mixed seizure types of Lennox–Gastaut syndrome, myoclonic, and generalized tonic clonic seizure. Recent studies have proved that zonisamide can acts as a migraine preventative medication and is effective in several cases of neuropathic pain. In an open-label trial, zonisamide shows positive effects on attenuating the symptoms of tardive dyskinesia.
It is assumed that zonisamide functions on the sodium and calcium channels in the brain cells, in which it controls electric-currents that are responsible for seizure activity. The FDA approved zonisamide in March 2000.