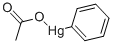
PHENYLMERCURIC ACETATE
- русский язык имя
- английское имяPHENYLMERCURIC ACETATE
- CAS №62-38-4
- CBNumberCB6703824
- ФормулаC8H8HgO2
- мольный вес336.74
- EINECS200-532-5
- номер MDLMFCD00008691
- файл Mol62-38-4.mol
| Температура плавления | 148-151 °C(lit.) |
| плотность | 2,4 g/cm3 |
| температура хранения | APPROX 4°C |
| растворимость | Slightly soluble in water, soluble in acetone and in alcohol. |
| форма | Powder |
| Удельный вес | 2.4 |
| цвет | white |
| Запах | Acetic acid odor |
| Растворимость в воде | Soluble in alcohol, benzene and glacial acetic acid. Slightly soluble in water. |
| Мерк | 14,7300 |
| Стабильность | Stable. Incompatible with strong oxidizing agents. |
| Справочник по базе данных CAS | 62-38-4(CAS DataBase Reference) |
| FDA 21 CFR | 310.545 |
| FDA UNII | OSX88361UX |
| Пестициды Закон о свободе информации (FOIA) | Phenylmercuric acetate |
| Система регистрации веществ EPA | Phenylmercury acetate (62-38-4) |
| UNSPSC Code | 12352103 |
| NACRES | NA.22 |
| Коды опасности | T,N | |||||||||
| Заявления о рисках | 25-34-48/24/25-50/53 | |||||||||
| Заявления о безопасности | 23-24/25-37-45-60-61 | |||||||||
| РИДАДР | UN 1674 6.1/PG 2 | |||||||||
| WGK Германия | 3 | |||||||||
| RTECS | OV6475000 | |||||||||
| F | 8 | |||||||||
| TSCA | Yes | |||||||||
| Класс опасности | 6.1 | |||||||||
| Группа упаковки | II | |||||||||
| кода HS | 28521000 | |||||||||
| Банк данных об опасных веществах | 62-38-4(Hazardous Substances Data) | |||||||||
| Токсичность | LD50 oral in rat: 41mg/kg | |||||||||
| NFPA 704: |
|
рисовальное письмо(GHS)
-
рисовальное письмо(GHS)




-
сигнальный язык
опасность
-
вредная бумага
H372:Поражает органы в результате многократного или продолжительного воздействия.
H314:При попадании на кожу и в глаза вызывает химические ожоги.
H410:Чрезвычайно токсично для водных организмов с долгосрочными последствиями.
H300:Смертельно при проглатывании.
-
оператор предупредительных мер
P260:Не вдыхать газ/ пары/ пыль/ аэрозоли/ дым/ туман.
P273:Избегать попадания в окружающую среду.
P280:Использовать перчатки/ средства защиты глаз/ лица.
P303+P361+P353:ПРИ ПОПАДАНИИ НА КОЖУ (или волосы): Снять/удалить немедленно всю загрязненную одежду. Промыть кожу водой.
P304+P340+P310:ПРИ ВДЫХАНИИ: Свежий воздух, покой. Немедленно обратиться за медицинской помощью.
P305+P351+P338:ПРИ ПОПАДАНИИ В ГЛАЗА: Осторожно промыть глаза водой в течение нескольких минут. Снять контактные линзы, если Вы ими пользуетесь и если это легко сделать. Продолжить промывание глаз.
PHENYLMERCURIC ACETATE химические свойства, назначение, производство
Химические свойства
Phenylmercuric acetate occurs as a white to creamy white, odorless or almost odorless, crystalline powder or as small white prisms or leaflets.Использование
Phenylmercuric acetate is used as catalyst; fungicide; herbicide; algicide; preservative in antibiotic eye drops, eye cosmetics, shampoos, etc.Методы производства
Phenylmercuric acetate is readily formed by heating benzene with mercuric acetate.Общее описание
Small lustrous prisms. Toxic by ingestion, inhalation and skin absorption. May severely irritate skin and eyes. Used as an herbicide and fungicide. as such, is mixed with organic solvent for the purpose of application.Профиль реактивности
PHENYLMERCURIC ACETATE may react with strong oxidizing agents .Угроза здоровью
Extremely toxic. The probable oral lethal dose for humans is 5-50 mg/kg, between 7 drops and 1 teaspoonful for a 70 kg (150 lb.) person.Пожароопасность
Fire may produce irritating or poisonous gases. When heated to decomposition, very toxic mercuric fumes may be given off. Phenylmercuric ion is incompatible with halides, with which precipitates are formed.Фармацевтические приложения
Phenylmercuric acetate is used as an alternative antimicrobial preservative to phenylmercuric borate or phenylmercuric nitrate in a limited range of cosmetics (in concentrations not exceeding 0.007% of mercury calculated as the metal) and pharmaceuticals. It may be used in preference to phenylmercuric nitrate owing to its greater solubility.Phenylmercuric acetate is also used as a spermicide;
Профиль безопасности
Poison by ingestion, intravenous, intraperitoneal, subcutaneous, and possibly other routes. An experimental teratogen. Other experimental reproductive effects. Mutation data reported. See also MERCURY COMPOUNDS. When heated to decomposition it emits toxic fumes of Hg.Безопасность
Phenylmercuric acetate is mainly used as an antimicrobial preservative in topical pharmaceutical formulations. A number of adverse reactions to mercury-containing preservatives have been reported; see Phenylmercuric Nitrate.LD50 (chicken, oral): 60 mg/kg
LD50 (mouse, IP): 13 mg/kg
LD50 (mouse, IV): 18 mg/kg
LD50 (mouse, oral): 13 mg/kg
LD50 (mouse, SC): 12 mg/kg
LD50 (rat, oral): 41 mg/kg
Возможный контакт
Phenylmercury acetate is used as an antiseptic, fungicide; for fungal and bacterial control; herbicide and control of crabgrass; mildewcide for paints; slimicide in paper mills. It was also used in contraceptive gels and foams.Экологическая судьба
If released into air, soil, or water, phenylmercuric acetate is unlikely to volatilize and is instead expected to be bound to particulates based on a low vapor pressure (6 × 10-6 mm Hg) and low Henry’s constant (5.66 × 10-10 atmm3 mol -1). Photolysis has the potential to degrade phenylmercuric acetate, releasing inorganic mercury which can volatilize and enter the atmosphere from superficial soils or water. If released into soil, the mobility of parent phenylmercuric acetic acid is expected to be high based on a Koc of 60. Water releases would result in quick dispersion since water solubility is high (4370 mg l-1). Once in solution, especially with harder water, it will dissociate into a salt. The cation will adsorb to particulates or humics suspended in the water column or in sediment, with little bioconcentration in aquatic species.хранилище
As for other phenylmercuric salts; see Phenylmercuric Nitrate.Phenylmercuric acetate should be stored in a well-closed container, protected from light, in a cool, dry place.
Перевозки
UN1674 Phenylmercuric acetate, Hazard Class: 6.1; Labels: 6.1-Poisonous materials.Методы очистки
It forms small colourless lustrous prisms from EtOH. Its solubility in H2O is 0.17%, but it is more soluble in EtOH, Me2CO and *C6H6. [Maynard J Am Chem Soc 46 1510 1925, Coleman et al. J Am Chem Soc 59 2703 1937, J Am Pharm Assoc 25 752 1936, Beilstein 16 IV 1720.] See PhHgOH below.Несовместимости
As for other phenylmercuric salts; see Phenylmercuric Nitrate.Incompatible with: halides; anionic emulsifying agents and suspending agents; tragacanth; starch; talc; sodium metabisulfite; sodium thiosulfate; disodium edetate; silicates; aluminum and other metals; amino acids; ammonia and ammonium salts; sulfur compounds; rubber; and some plastics.
Phenylmercuric acetate is reported to be incompatible with cefuroxime and ceftazidime.
Утилизация отходов
Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform with EPA regulations governing storage, transportation, treatment, and waste disposal. React to produce soluble nitrate form, precipitate as mercuric sulfide. Return to supplier.Регуляторный статус
Included in the FDA Inactive Ingredients Database (ophthalmic ointments; topical emulsions/creams; vaginal emulsions/creams). Included in the Canadian List of Acceptable Non-medicinal Ingredients (ophthalmic, nasal and otic preparations up to 0.004%; there must be no other suitable alternative preservative available).Phenylmercuric acetate is no longer permitted to be used as a pesticide in the USA. Its use in cosmetic products in the USA is limited to eye area cosmetics at not more than 0.0065% provided that there is no other suitable available preservative. It is specifically prohibited in vaginal contraceptive drug products and antimicrobial diaper rash drug products in the USA. Phenylmercuric compounds are prohibited from use in cosmetic products in Canada.
In Europe, use in cosmetic products is limited to eye makeup and eye makeup remover at concentrations not exceeding 0.007% mercury alone or in combination with other permitted mercurial compounds.In France, a maximum concentration of 0.01% is permitted for use in pharmaceuticals. The use of mercurial compounds in cosmetics in Japan is limited to concentrated shampoo or cream at not more than 0.003% Hg and eye makeup at not more than 0.0065% Hg.
PHENYLMERCURIC ACETATE запасные части и сырье
сырьё
1of2
запасной предмет