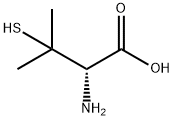
D(-)-пеницилламин
- английское имяD-Penicillamine
- CAS №52-67-5
- CBNumberCB5742367
- ФормулаC5H11NO2S
- мольный вес149.21
- EINECS200-148-8
- номер MDLMFCD00064302
- файл Mol52-67-5.mol
| Температура плавления | 210 °C (dec.)(lit.) |
| Температура кипения | 251.8±35.0 °C(Predicted) |
| альфа | -65 º (c=5, 1M NaOH, on dry) |
| плотность | 1.113 (estimate) |
| показатель преломления | -63 ° (C=1, 1mol/L NaOH) |
| температура хранения | 2-8°C |
| растворимость | H2O: soluble100mg/mL |
| форма | Powder |
| пка | pKa 7.83±0.01(H2O,t =37±0.05,I=0.15)(Approximate) |
| цвет | White to almost white |
| Растворимость в воде | 11.1 g/100 mL (20 ºC) |
| Мерк | 14,7088 |
| БРН | 1722375 |
| BCS Class | 3 |
| Стабильность | Stable. Incompatible with strong oxidizing agents. |
| ИнЧИКей | VVNCNSJFMMFHPL-VKHMYHEASA-N |
| Справочник по базе данных CAS | 52-67-5(CAS DataBase Reference) |
| Рейтинг продуктов питания EWG | 1 |
| Словарь онкологических терминов NCI | penicillamine |
| FDA UNII | GNN1DV99GX |
| Словарь наркотиков NCI | Cuprenil |
| Код УВД | M01CC01 |
| Предложение 65 Список | Penicillamine |
| Справочник по химии NIST | Penicillamine(52-67-5) |
| Система регистрации веществ EPA | Penicillamine (52-67-5) |
| Коды опасности | Xi,T,Xn | |||||||||
| Заявления о рисках | 36/37/38-40-20/21/22 | |||||||||
| Заявления о безопасности | 26-36-24/25-22 | |||||||||
| WGK Германия | 2 | |||||||||
| RTECS | YV9425000 | |||||||||
| Примечание об опасности | Toxic | |||||||||
| кода HS | 29309016 | |||||||||
| Банк данных об опасных веществах | 52-67-5(Hazardous Substances Data) | |||||||||
| Токсичность | LD50 in rats (mg/kg): >10000 orally, >660 i.p. (Jaffe) | |||||||||
| NFPA 704: |
|
рисовальное письмо(GHS)
-
рисовальное письмо(GHS)

-
сигнальный язык
предупреждение
-
вредная бумага
H361d:Предполагается, что данное вещество может отрицательно повлиять на неродившегося ребенка.
-
оператор предупредительных мер
P201:Беречь от тепла, горячих поверхностей, искр, открытого огня и других источников воспламенения. Не курить.
P202:Перед использованием ознакомиться с инструкциями по технике безопасности.
P280:Использовать перчатки/ средства защиты глаз/ лица.
P308+P313:ПРИ подозрении на возможность воздействия обратиться за медицинской помощью.
P405:Хранить в недоступном для посторонних месте.
P501:Удалить содержимое/ контейнер на утвержденных станциях утилизации отходов.
D(-)-пеницилламин химические свойства, назначение, производство
Описание
Penicillamine is an orally bioavailable copper chelator and penicillin degradation product. It increases urinary and fecal copper excretion and decreases liver copper concentration in a rat model of copper overload when administered at 0.67 mmol/kg per day, but does not affect kidney, spleen, or brain copper levels. Penicillamine (100 mg/kg per day) dissolves copper-rich granules in hepatic lysosomes of Long-Evans cinnamon (LEC) rats, which spontaneously develop hepatic injury and acute hepatitis and have a mutation homologous to that of the human Wilson disease gene. Penicillamine has anticonvulsant and proconvulsant effects in mice when administered at 0.5 and 250 mg/kg, respectively, which are blocked by the nitric oxide synthase (NOS) inhibitors L-NAME and 7-nitroindazole . Formulations containing penicillamine have been used to treat Wilson disease, cystinuria, and active rheumatoid arthritis.Химические свойства
White to off-white crystalline powderИспользование
As a Penicillin metabolite, D-(-)-Penicillamin can be used in the treatment of Wilson’s disease, Cystinuria, Scleroderma and arsenic poisoning.Определение
ChEBI: An optically active form of penicillamine having D-configuration. Pharmaceutical form (L-form is toxic) of chelating agent used to treat heavy metal poisoning.Общее описание
D-Penicillamine contains a β-lactam chemical structure.Профиль безопасности
Poison by intraperitoneal route. Moderately toxic by subcutaneous and intravenous routes. Mildly toxic by ingestion. An experimental teratogen. Human systemic effects by ingestion: agranulocytosis, dermatitis, fever, hemorrhage, increased body temperature, dermatitis, leukopenia, proteinuria, thrombocytopenia. Human teratogenic effects by an unspecified route: developmental abnormalities of the craniofacial areas, skin, and skin appendages, and body wall. Experimental reproductive effects. Questionable human carcinogen producing leukemia. Mutation data reported. Used in the treatment of rheumatoid arthritis, metal poisonings, and cystinuria. When heated to decomposition it emits very toxic fumes of NOx and SOx. See also MERCAPTANS.Синтез
d-penicillamine can be synthesized in a multistep process that begins with heating isobutyraldehyde, pyridine, sulfur, and ammonia in benzene to form 5,5-dimethyl-2- isopropyl-?3-thiazoline. Treatment with hydrogen cyanide gives 4-cyano-5,5-dimethyl- 2-isopropylthiazolidine, which on acid hydrolysis gives d,l-penicillamine hydrochloride. Resolution is accomplished by conversion of the racemate to d,l-3-formyl- 2,2,5,5-tetramethylthiazolidine-4-carboxylic acid by treatment first with acetone, then with acetic formic anhydride. The enantiomers are separated in the usual manner, using, for example, l-lysine or d-(?)- threo-1-(4-nitrophenyl)-2-aminopropane-1,3- diol. Acidification liberates d-3-formyl- 2,2,5,5-tetramethylthiazolidine-4-carboxylic acid, which is hydrolyzed with hydrochloric acid to yield d-penicillamine hydrochloride. Neutralization with ethanolic triethylamine affords d-penicillamine.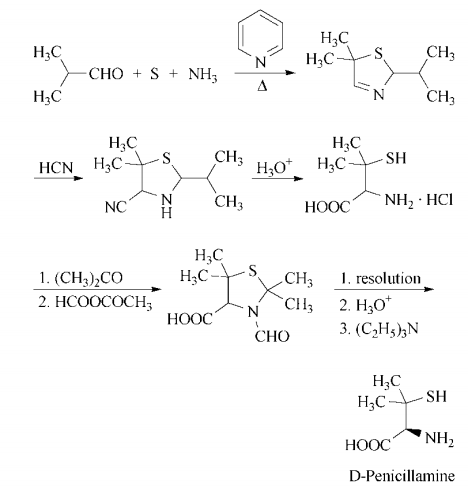
Методы очистки
The melting point of D-(-)-penicillamine depends on the rate of heating (m 202-206o is obtained by starting at 195o and heating at 2o/minute). It is soluble in H2O and alcohols but insoluble in Et2O, CHCl3, CCl4 and hydrocarbon solvents. Purify it by dissolving it in MeOH and adding Et2O slowly. Dry it in vacuo and store it under N2. [Weight et al. Angew Chem, Int Ed (English) 14 330 1975, Cornforth in The Chemistry of Penicillin (Clarke, Johnson and Robinson eds) Princeton Univ Press, 455 1949, Review: Chain et al. Antibiotics (Oxford University Press) 2 1949, Polymorphism: Vidler J Pharm Pharmacol 28 663 1976]. The D-S-benzyl derivative has m 197-198o (from H2O), [] D 17 -20o (c 1, N NaOH), -70o (N HCl). [Beilstein 4 IV 3228.]D(-)-пеницилламин запасные части и сырье
сырьё
1of2
запасной предмет
D(-)-пеницилламин поставщик
| поставщик | телефон | страна | номенклатура продукции | благоприятные условия |
|---|---|---|---|---|
| 571-85586718 +8613336195806 |
China | 29798 | 60 | |
| +86-371-66670886 | China | 16216 | 58 | |
| 010-60279497 | CHINA | 1811 | 55 | |
| +86-0371-55170693 +86-19937530512 |
China | 21670 | 55 | |
| 008657128800458; +8615858145714 |
China | 9337 | 55 | |
| +86-0371-86658258 +8613203830695 |
China | 29897 | 58 | |
| +86-15013270415 | China | 246 | 58 | |
| +86 18953170293 | China | 2931 | 58 | |
| +8615866703830 | China | 7353 | 58 | |
| 18631714998 | CHINA | 906 | 58 |