ХЛОРИД БЕРИЛЛИЯ
- английское имяberyllium chloride
- CAS №7787-47-5
- CBNumberCB1279478
- ФормулаBeCl2
- мольный вес79.92
- EINECS232-116-4
- номер MDLMFCD00042674
- файл Mol7787-47-5.mol
| Температура плавления | 399 °C(lit.) | ||||||||||||||
| Температура кипения | 520°C | ||||||||||||||
| плотность | 1.899 g/mL at 25 °C(lit.) | ||||||||||||||
| плотность пара | 1 mm at 291°C (subl) | ||||||||||||||
| растворимость | soluble in ethanol, ethyl ether, pyridine; insoluble in benzene, toluene | ||||||||||||||
| форма | white-yellow orthorhombic crystals | ||||||||||||||
| цвет | Colorless, deliquescent needles, or orthorhombiccrystals | ||||||||||||||
| Растворимость в воде | very soluble H2O with evolution of heat [MER06]: g/100g solution H2O: 40.35 (0°C), 41.72 (25°C); solid phase, BeCl2 · 4H2O [KRU93]; soluble alcohol, benzene, ether [HAW93] | ||||||||||||||
| Мерк | 13,1170 | ||||||||||||||
| crystal system | square | ||||||||||||||
| Space group | I41/acd | ||||||||||||||
| Lattice constant |
|
||||||||||||||
| Справочник по базе данных CAS | 7787-47-5 | ||||||||||||||
| Рейтинг продуктов питания EWG | 3 | ||||||||||||||
| FDA UNII | 58B7024067 | ||||||||||||||
| Система регистрации веществ EPA | Beryllium chloride (7787-47-5) |
| Коды опасности | T+,N |
| Заявления о рисках | 49-25-26-36/37/38-43-48/23-51/53 |
| Заявления о безопасности | 53-45-61 |
| РИДАДР | UN 1566 6.1/PG 2 |
| WGK Германия | 3 |
| RTECS | DS2625000 |
| F | 3-10 |
| Класс опасности | 6.1(b) |
| Группа упаковки | III |
| Банк данных об опасных веществах | 7787-47-5(Hazardous Substances Data) |
| Токсичность | LD50 orl-rat: 86 mg/kg HYSAAV 30,169,65 |
рисовальное письмо(GHS)
-
рисовальное письмо(GHS)

-
сигнальный язык
предупреждение
-
вредная бумага
H315:При попадании на кожу вызывает раздражение.
H319:При попадании в глаза вызывает выраженное раздражение.
-
оператор предупредительных мер
P264:После работы тщательно вымыть кожу.
P280:Использовать перчатки/ средства защиты глаз/ лица.
P302+P352:ПРИ ПОПАДАНИИ НА КОЖУ: Промыть большим количеством воды.
P305+P351+P338:ПРИ ПОПАДАНИИ В ГЛАЗА: Осторожно промыть глаза водой в течение нескольких минут. Снять контактные линзы, если Вы ими пользуетесь и если это легко сделать. Продолжить промывание глаз.
P332+P313:При возникновении раздражения кожи: обратиться за медицинской помощью.
P337+P313:Если раздражение глаз не проходит обратиться за медицинской помощью.
P362+P364:Снять всю загрязненную одежду и выстирать ее перед повторным использованием.
ХЛОРИД БЕРИЛЛИЯ химические свойства, назначение, производство
Химические свойства
Beryllium chloride is available as colorless to yellow crystals. It decomposes rapidly on contact with water producing hydrogen chloride, and attacks many metals in the presence of water. Beryllium chloride emits irritating or toxic fumes (or gases) in a fi re.Физические свойства
Beryllium chloride has the formula BeCl2 with a molecular weight of 79.9176 g/mol. Beryllium chloride, BeCl2, melts at 405°C and boils at 520°C. That compares with 714°C and 1412°C for magnesium chloride. The solid is a one-dimensional polymer consisting of edge-shared tetrahedra. In contrast, BeF2 is a threedimensional polymer, with a structure akin to that of quartz. In the gas phase, BeCl2 exists both as a linear monomer and a bridged dimer with two bridging chlorine atoms where the beryllium atom is 3-coordinate. This linear shape contrasts with the monomeric forms of some of the dihalides of the heavier members of group 2, e.g. CaF2, SrF2, BaF2, SrCl2, BaCl2 and Ba that are all nonlinear.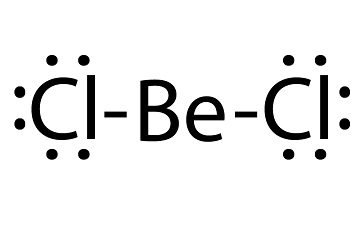
beryllium chloride lewis structure
The pure chloride is a glassy, transparent mass having a specific gravity of 2.01 at 15°C. It becomes fluid toward 440°C, passing through a viscous condition, but above 800° C it begins to volatilize, yielding white and very deliquescent crystals. It dissolves readily in water, but is only slightly soluble in absolute alcohol. By cooling an alcoholic solution to 23°C, one can obtain a white crystalline mass which, however, melts easily as the temperature rises.
Beryllium chloride, an electron-deficient compound similar to aluminum chloride, is a Lewis acid. The anhydrous salt is used as a catalyst in organic reactions.
История
Although early workers (1798) obtained the chloride in solution, the pure salt was not made until about 1827 when one worker of the time prepared it in the sublimed anhydrous state by passing chlorine gas over a heated mixture of carbon and beryllium oxide. Finally (1885), it was prepared as the pure chloride in very pure form for the purpose of determining its vapor density by the action of dry hydrochloric acid gas on the metal, but the pure salt was not produced until 1898 when one worker made it by heating the double fluoride of ammonium and beryllium, which had previously been dried over phosphoric anhydride, in a current of dry carbon dioxide and cooled in an atmosphere of the same gas. He also prepared it by the action of hydrofluoric acid gas on the carbide. No matter what method is used, it was determined that the materials must be absolutely dry if a pure chloride is to be obtained.Использование
Beryllium chloride (BeCl2) is used as a catalyst to accelerate many organic reactions, and beryllium chloride is the electrolyte used along with NaCl in the electrolytic process to produce beryllium metal.Подготовка
Beryllium chloride is prepared by passing chlorine over beryllium oxide and carbon:BeO + C + Cl2 → BeCl2 + CO
It also is made by combination of beryllium with chlorine.
Определение
ChEBI: A compound of beryllium (+2 oxidation state) and chloride in the ratio 1:2.Общее описание
White to green solid with a sharp odor.Реакции воздуха и воды
Reacts with water with evolution of heat. Forms beryllium oxide and hydrochloric acid solution. Corrodes most metals in presence of moisture. Flammable and explosive hydrogen gas may collect in enclosed spaces [USCG, 1999].Профиль реактивности
Acidic salts, such as BERYLLIUM CHLORIDE, are generally soluble in water. The resulting solutions contain moderate concentrations of hydrogen ions and have pH's of less than 7.0. They react as acids to neutralize bases. These neutralizations generate heat, but less or far less than is generated by neutralization of inorganic acids, inorganic oxoacids, and carboxylic acid. They usually do not react as either oxidizing agents or reducing agents but such behavior is not impossible. Many of these compounds catalyze organic reactions. BERYLLIUM CHLORIDE reacts with vigor with sulfur nitrides. Some explode upon intimate mixing, i.e. tetrasulfur tetranitride.Опасность
Very toxic.Угроза здоровью
Exposures to beryllium chloride cause redness, pain and blurred vision, nausea, vomiting, and abdominal pain. Inhalation of beryllium chloride causes cough, sore throat, shortness of breathПожароопасность
Special Hazards of Combustion Products: Toxic and irritating beryllium oxide fumes and hydrogen chloride may form in fires.Профиль безопасности
Confirmed carcinogen withexperimental tumorigenic data. Poison by ingestion andintraperitoneal routes. An experimental teratogen. Otherexperimental reproductive effects. Mutation data reported.When heated to decomposition it emits very toxic fumesofСинтез
Beryllium chloride can be prepared directly from beryl by chloride or by chlorination of beryllium oxide under reducing conditions. Beryllium chloride is especially well suited for purification by distillation in a stream of hydrogen and fractional condensation. The significantly lower-boiling chlorides of aluminum, silicon, and iron(III) can be separated by careful temperature control. Iron(II) chloride, which is reduced by hydrogen, stays in the residue.ХЛОРИД БЕРИЛЛИЯ запасные части и сырье
сырьё
ХЛОРИД БЕРИЛЛИЯ поставщик
| поставщик | телефон | страна | номенклатура продукции | благоприятные условия |
|---|---|---|---|---|
| 18871490254 | CHINA | 28172 | 58 | |
| 86-13657291602 | CHINA | 22963 | 58 | |
| +86-023-6139-8061 +86-86-13650506873 |
China | 39894 | 58 | |
| 18503026267 | CHINA | 9636 | 58 | |
| +8615604608665 15604608665 |
CHINA | 9414 | 58 | |
| +86-89586680 +86-13289823923 |
China | 8670 | 58 | |
| +86-85511178; +86-85511178; |
China | 35425 | 58 | |
| +86-852-30606658 | China | 43340 | 58 | |
| +86-0592-6210733 | China | 32343 | 55 | |
| 010-56205725 | China | 12335 | 58 |