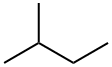
What is Isopentane?
Aug 12,2020
General Description
Isopentane is an alkane that is butane substituted by a methyl group at position 2. Isopentane is an extremely volatile and extremely flammable liquid at room temperature and pressure. It is also the least dense liquid at standard conditions.[citation needed] The normal boiling point is just a few degrees above room temperature and isopentane will readily boil and evaporate away on a warm day.
Isopentane is commonly used in conjunction with liquid nitrogen to achieve a liquid bath temperature of −160 °C. Natural gas typically contains 1% or less isopentane, but it is a significant component of natural gasoline. Isopentane is a low molecular-weight alkane derived from the petroleum raw materials natural gas and crude oil. It is a flammable liquid and has physical properties very similar to those of n-pentane. It is relatively stable and is inert to most chemical agents. lsopentane is generally marketed in a compressed liquefied form. Isopentane is used as a solvent, is a building-block chemical used in reactions to form other industrial substances, and has been used in the manufacture of chlorinated derivatives and the production of amyl-naphthalene and isoprene. It is used as an aerosol propellant industrially and as a blowing agent for polystyrene.
Structure & Properties
2-Methylbutane (isopentane), C5H12, is a flammable liquid and exhibits physical properties very similar to those of pentane. It has been detected in urban air.2-Methylbutane is an alkane that is butane substituted by a methyl group at position 2. It has a higher BP than butane because, although similarly branched, it has a higher MW. A useful analogy for comparing molecules with the same size of longest chain is that of cylindrical-shaped molecules. It has a role as a refrigerant. Biological samples flash frozen for example with a combination of liquid nitrogen and methylbutane can then be used for storage, cryosection, etc.
Watery colorless liquid with a gasoline-like odor. Floats on water. Flammable, irritating vapor is produced. Boiling point is 82°F.
Uses
Isopentane is an organic, branched-chain alkane with five carbon atoms. 2-Methylbutane undergoes catalytic dehydrogenation in the presence of chromia-alumina catalyst to form isoamylenes, which can undergo further dehydrogenation to form isoprene. It may also be used as a solvent in the preparation of trans-Bis(triethylphosphine) (hydroxy carbonyl) (phenyl) platinum(II), a metallacarboxylic acid. It can be used as a solvent, in manufacture of chlorinated derivatives, and blowing agent for polystyrene.
Isopentane is used in a closed loop in geothermal power production to drive turbines, used in conjunction with dry ice or liquid nitrogen, to freeze tissues for cryosectioning in histology.
Isopentane is a major component (sometimes 30% or more) of natural gasoline, an analog of common petroleum-derived gasoline that is condensed from natural gas. It has a substantially higher octane rating (RON 93.7) than n-pentane (61.7), and therefore there is interest in conversion from the latter.
Because of the use of isopentane and other light aliphatic paraffinic hydrocarbons in cosmetic products, an independent "blue ribbon" panel of experts published in the peer-reviewed literature a review paper on the health and safety aspects of their cosmetic uses. The publication is the Final Report of the Safety Assessment of lsobutane, Isopentane, n-Butane, and Propane (Expert Panel, 1982). The panel concluded that "on the basis of the available information, isopentane, isobutane, n-butane and propane are considered safe as cosmetic ingredients under present conditions of concentration and use.
Environmental fate Photolytic. When synthetic air containing gaseous nitrous acid and 2-methylbutane was exposed to artificial sunlight (λ = 300–450 nm), acetone, acetaldehyde, methyl nitrate, peroxy-acetal nitrate, propyl nitrate, and pentyl nitrate were formed (Cox et al., 1980).
Based upon a photooxidation rate constant of 3.90 x 10-12 cm3/molecule with OH radicals in summer daylight, the atmospheric lifetime is 36 h (Altshuller, 1991). At atmospheric pressure and 300 K, Darnall et al. (1978) reported a rate constant of 3.78 x 10-12 cm3/molecule for the same reaction.
Cox et al. (1980) reported a rate constant of 5.0 x 10-11 cm3/molecule for the reaction of gaseous 2-methylbutane with OH radicals based on a value of 8 x 10-12 cm3/molecule?sec for the reaction of ethylene with OH radicals.
Chemical/Physical. Complete combustion in air produces carbon dioxide and water vapor.
2-Methylbutane will not hydrolyze because it does not contain a hydrolyzable functional group.
Safety Profile
lsopentane is practically nontoxic. Breathing low concentrations has no effect; very high concentrations causes drowsiness to anesthesia, depending on the concentration. Only very high vapor concentrations of isopentane produce alterations in normal body functions. No permanent alterations in body function are induced by isopentane. Eliminating exposure to vapors allows the body to completely recover to normal.
The toxic properties of isopentane are not manifest until levels are reached that exceed the minimum fire hazard concentrations. The lower flammability limit of isopentane is 1.4 vol% in air, which is 14,000 ppm.
You may like
Exploring the Compound H2O: Water
Feb 27, 2024
Related articles And Qustion
Lastest Price from 2-Methylbutane manufacturers
2-Methylbutane

US $10.00/KG2025-04-21
- CAS:
- 78-78-4
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 100 mt
2-Methylbutane

US $23.00/L2025-01-16
- CAS:
- 78-78-4
- Min. Order:
- 1L
- Purity:
- 0.99
- Supply Ability:
- 3000000tons