What are the effects of Pyridine on human health and the environment?
Description of Pyridine
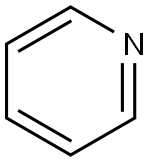
Pyridine is a flammable, colourless liquid, six-membered heterocyclic compound with an unpleasant odour. Pyridine has a variety of uses in organic chemistry and industrial practice, including as an important solvent, reagent and precursor in pesticides and pharmaceuticals. Pyridine derivatives are considered a promising antiviral drug due to increasing drug resistance. Pyridine was first synthesised from acetylene and hydrogen cyanide, and the reaction of acetaldehyde and formaldehyde with ammonia is the most widely used method of pyridine production. Pyridine can also be formed by the decomposition of many natural substances in the environment.

Health Effects
Pyridine can cause first degree burns when exposed for short periods of time. The most important health problem for humans exposed to pyridine would be liver damage. Other health problems in humans may be neurological effects, kidney effects, and skin and eye irritation. Pyridine is a confirmed carcinogen in animals, but it is not known to cause cancer, birth defects or reproductive problems in humans and may be carcinogenic to humans.
Pyridine derivatives can be divided into two main groups: pyridine-containing heterocycles and pyridine-fused rings. Studies have shown that pyridine scaffold derivatives possess a wide range of biological activities such as antimicrobial, antiviral, antioxidant, antidiabetic, anticancer, antimalarial, analgesic and anti-inflammatory. In addition, they have psychopharmacological, antagonistic, anti-amoebic and antithrombotic activities. These derivatives possess good antiviral activity against different types of viruses such as human immunodeficiency virus (HIV), hepatitis C virus (HCV), hepatitis B virus (HBV), respiratory syncytial virus (RSV) and cytomegalovirus (CMV). These derivatives inhibit viral applications through different mechanisms of action such as RT inhibition, polymerase inhibition, RNase H activity inhibition, maturation inhibition, viral thymidine kinase inhibition, AAK1 (Adaptor-Associated kinase 1) inhibition, GAK (Cyclin G-associated kinase) inhibition, post-integration event inhibition, HDAC6 inhibition, CCR5 antagonist activity inhibition, DNA and RNA replication inhibition, gene expression inhibition, cellular NF-jB signalling pathway and neuraminidase (NA) inhibition, protein synthesis inhibition, and general inhibition of the viral replication cycle.
Environment Effects
Pyridine is present in the environment as a by-product of coal gasification and can easily evaporate into the air. If pyridine is released into the air, it can take months to years to break down into other compounds. Pyridine also mixes easily with water. If it is released into water or soil, it may break down within days to months.
Pyridine is a widely employed nitrogen-containing heterocyclic organic, and the discharge of pyridine wastewater poses substantial environmental challenges due to its recalcitrance and toxicity. Co-metabolic degradation emerged as a promising solution. The studies used easily degradable glucose and structurally similar phenol as co-metabolised substrates, respectively, and delved into the corresponding mechanisms. To treat 400 mg/L pyridine, all reactors achieved remarkably high removal efficiencies, surpassing 98.5%. And the co-metabolism reactors had much better pyridine-N removal performance. Batch experiments revealed that glucose supplementation bolstered nitrogen assimilation, thereby promoting the breakdown of pyridine, and resulting in the highest pyridine removal rate and pyridine-N removal efficiency. The high abundance of Saccharibacteria (15.54%) and the enrichment of GLU and glnA substantiated this finding. On the contrary, phenol delayed pyridine oxidation, potentially due to its higher affinity for phenol hydroxylase. Nevertheless, phenol proved valuable as a carbon source for denitrification, augmenting the elimination of pyridine-N. This was underscored by the abundant Thauera (30.77%) and Parcubacteria (7.21%) and the enriched denitrification enzymes. This study demonstrated that co-metabolic degradation can bolster the simultaneous conversion of pyridine and pyridine-N, and shed light on the underling mechanism.
Conventional pyridine degradation systems are inefficient and have poor nitrogen removal. The study elucidated the simultaneous removal performance of pyridine and pyridine-N with the addition of glucose or phenol and demonstrated the synergistic metabolic mechanism. Glucose accelerated the oxidation of pyridine and improved the TN removal by enhancing ammonia assimilation. Phenol, although removing TN by denitrification, hindered the degradation of pyridine. This study provides important information for the effective removal of carbon and nitrogen from high-strength pyridine wastewater and sheds light on treatment methods for difficult-to-degrade nitrogenous organic wastewater.
References:
[1] SEYEDEH ROYA ALIZADEH; Mohammad A E. Antiviral Activities of Pyridine Fused and Pyridine Containing Heterocycles, A Review (from 2000 to 2020).[J]. Mini reviews in medicinal chemistry, 2021. DOI:10.2174/1389557521666210126143558.
[2] FAN Y, YAN D, CHEN X, et al. Novel insights into the co-metabolism of pyridine with different carbon substrates: performance, metabolism pathway and microbial community[J]. Journal of Hazardous Materials, 2023. DOI:10.1016/j.jhazmat.2023.133396.
Related articles And Qustion
See also
Lastest Price from Pyridine manufacturers
US $1.00/kg2025-04-21
- CAS:
- 110-86-1
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 10 mt
US $1.10/g2025-04-17
- CAS:
- 110-86-1
- Min. Order:
- 1g
- Purity:
- 99.0% min
- Supply Ability:
- 100 tons min