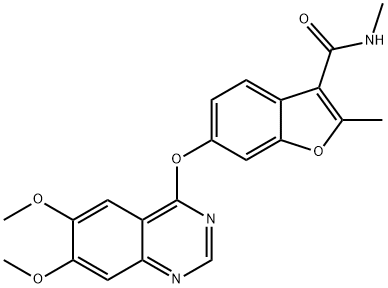
The pharmacodynamics and Synthetic method of Fruquintinib
Description
Fruquintinib, a highly potent and selective vascular endothelial growth factor (VEGFR)-1, -2, and -3 tyrosine kinase inhibitor disclosed by Hutchinson MediPharma, was approved in China in 2018 for treating solid tumors. The drug was approved for the treatment of metastatic colorectal cancer for patients who had at least two prior failed treatments involving systemic antineoplastic therapies. Several additional clinical trials are currently underway evaluating fruquintnib for the treatment of advanced gastric cancer and advanced non-small-cell lung cancer.
Pharmacodynamics
Fruquintinib is a potent small-molecule tyrosine kinase inhibitor (TKI) with high VEGFR-1, -2, and -3 selectivity. In an in vitro evaluation of fruquintinib against a panel of 253 kinases, fruquintinib inhibited VEGFR-1, -2, and -3 with IC50 values of 33, 35, and 0.5 nmol/L, respectively; it had weak activity (IC50 values of 128–458 nmol/L) against RET, FGFR-1 and c-kit kinases. However, it had no significant inhibitory effect on any other kinase tested (IC50 values all > 1000 nmol/L)[1].
In vitro cellular assays, fruquintinib suppressed VEGF/VEGFR signaling and cell proliferation. Furthermore, the anti-angiogenic activity of fruquintinib was demonstrated in in vitro and ex vivo assays and in a tumor xenograft model.
In vivo, fruquintinib inhibited tumor growth in multiple human tumor xenograft murine models, including xenograft models of gastric, colon, and lung cancer. Of note, an enhanced antitumor effect was observed when fruquintinib was administered with chemotherapy agents (docetaxel or oxaliplatin). An antitumor synergistic impact has also been observed in xenograft models when fruquintinib is combined with other targeted therapies, including the EGFR TKIs gefitinib and the imatinib, and the c-MET inhibitor ruxolitinib. Also of note, the co-administration of fruquintinib and an anti-PD-L1 antibody provided an enhanced antitumor effect in a syngeneic murine tumor model compared with either agent alone.
Synthetic method
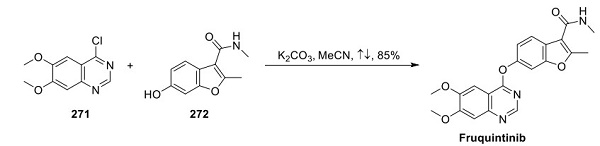
Although a discovery or process chemistry route for the synthesis of fruquintinib has yet to be disclosed, several U.S. and Chinese patents have been filed relating to the isolation of various crystalline forms of the drug[2]. A generic reaction for the final transformation was reported in two patents, which involves the SNAr reaction between chloroquinazoline 271 and phenol 272, affording fruquintinib an 85% yield, as shown above.
References
[1] Shirley, Matt. “Fruquintinib: First Global Approval.” Drugs 78 16 (2018): 1757–1761.
[2] Andrew C. Flick. “Synthetic Approaches to New Drugs Approved during 2018.” Journal of Medicinal Chemistry 63 19 (2020): 10652–10704
You may like
Related articles And Qustion
See also
Lastest Price from Fruquintinib manufacturers

US $0.00-0.00/kg2025-04-21
- CAS:
- 1194506-26-7
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- 20tons

US $0.00-0.00/g2025-04-21
- CAS:
- 1194506-26-7
- Min. Order:
- 2g
- Purity:
- 99%
- Supply Ability:
- 1000kg