Synthesis and Clinical study of Upadacitinib
General description
Upadacitinib is an oral JAK1 inhibitor used to treat rheumatoid arthritis, psoriatic arthritis and other diseases. JAK kinases are a family of non-receptor tyrosine kinases. Four members have been identified, namely JAK1, JAK2, JAK3 and TYK1. The substrate of JAK is STAT, a signal transducer and activator of transcription. STAT dimerizes after being phosphorylated by JAK, and then crosses the nuclear membrane into the nucleus to regulate the expression of related genes. This signaling pathway is called JAK-STAT pathway. Therefore, JAK plays an important role in the pathophysiology of immune-mediated diseases, and can be used to treat some autoimmune diseases, such as atopic dermatitis and rheumatoid arthritis. Psoriasis, ulcerative colitis, etc.

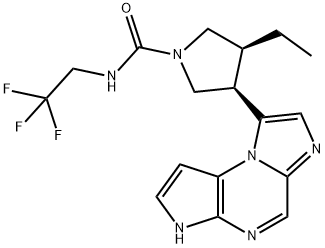
Fig. 1 The structure of Upadacitinib.
Synthetic routes
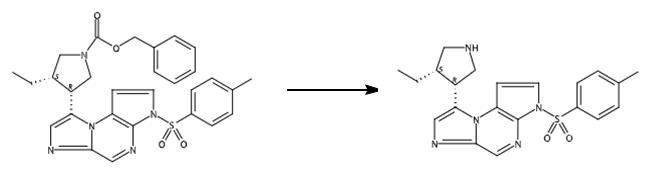
Fig. 2 The synthetic step 1 of Upadacitinib.
To a solution of (cis)-benzyl 3-ethyl-4-(3-tosyl-3H-imidazo[1,2-a]pyrrolo[2,3-e]pyrazin-8-yl)pyrrolidine-1-carboxylate (0.838 g, 1.541 mmol) was added a solution of HBr (2.50 mL, 15.19 mmol, 33% in acetic acid). The reaction mixture was stirred at ambient temperature for about 1 h. The reaction was diluted with Et2O (50 mL) and water (20 mL). The layers were stirred for about 3 min and the organic layer was decanted then the procedure was repeated 5 times. The aqueous layer was cooled to about 0 °C was basified with saturated aqueous NaHCO3 solution (10 mL) to about pH 7. The aqueous layer was extracted with EtOAc (3 x 50 mL), combined, and dried over anhydrous Na2SO4, filtered and concd to give a brown solid. The solid was dissolved in DCM (50 mL) and washed with water (3 x 20 mL), dried over anhydrous Na2SO4, filtered and concd to afford 8-((cis)-4-ethylpyrrolidin-3-yl)-3-tosyl-3H-imidazo[1,2-a]pyrrolo[2,3-e]pyrazine. Brown residue, yield 0.453, 61%. LC/MS (Table 1, Method a) Rt = 1.73 min; MS m/z: 410 (M+H)+ [1].
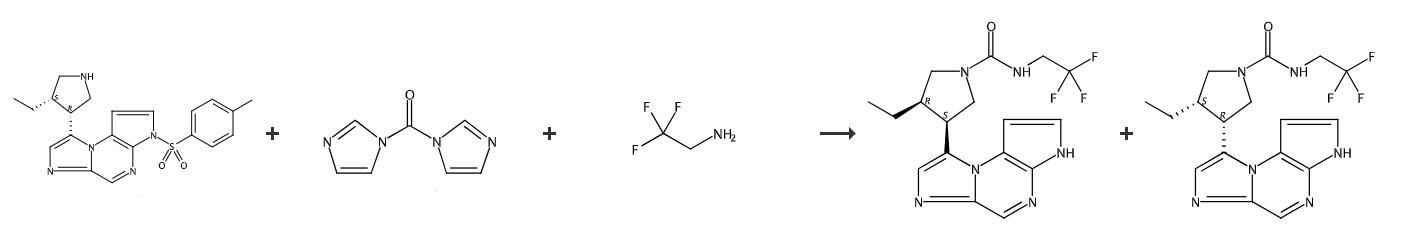
Fig. 3 The synthetic step 2 of Upadacitinib.
To a slurry of (5-tosyl-5H-pyrrolo[2,3-b]pyrazin-2-yl)methanamine hydrochloride (0.50 g, 1.5 mmol) in DCM (10 mL) at about 0 °C was added TEA (0.226 mL, 1.62 mmol). To the homogeneous reaction mixture was added a solution of 1,1'-thiocarbonyldiimidazole (0.29 g, 1.6 mmol) in DCM (10 mL). After about 30 min, a slurry of tert-butyl pyrrolidin-3-ylcarbamate (0.83 g, 4.4 mmol, TCI) in DCM (10 mL) was added to the reaction mixture. After stirring for about 20 min, the reaction mixture was allowed to warm to ambient temperature. After stirring for about 15 h, saturated aqueous NaHCO3 (30 mL) was added to the reaction mixture. The organic layer was separated, concd in vacuo, and purified by chromatography on silica gel eluting with 20-40% EtOAc in DCM to provide tert-butyl 1-((5-tosyl-5H-pyrrolo[2,3-b]pyrazin-2-yl)methylcarbamothioyl)pyrrolidin-3-ylcarbamate. Preparation #D.1*: (3R,4R)-tert-butyl-3-(6H-imidazo[1,5-a]pyrrolo[2,3-e]pyrazin-1-yl)-4-methylpiperidine-1-carboxylate To a solution of (3R,4R)-tert-butyl 4-methyl-3-(6-tosyl-6H-imidazo[1,5-a]pyrrolo[2,3-e]pyrazin-1-yl)piperidine-1-carboxylate (40 g, 78 mmol, Example #5 Step H) in 1,4-dioxane (160 mL) was added NaOH (1 N aqueous, 157 mL). The reaction was heated at about 60 °C for about 1 h. The reaction was allowed to cool to ambient temperature. The reaction was neutralized with aqueous HCl (4 N, 50 mL). The layers and extracted with DCM (2 x 300 mL). The combined organic extracts were washed with brine (400 mL), dried over anhydrous Na2SO4, filtered then concd in vacuo. The product was purified by chromatography on silica gel (330 g) using 1-5% MeOH in DCM to give (3R,4R)-tert-butyl 3-(6H-imidazo[1,5-a]pyrrolo[2,3-e]pyrazin-1-yl)-4-methylpiperidine-1-carboxylate. Final product AA.1.159:Rt min (method): 1.52 (a); m/z ESI+ (M+H)+: 381. AA.1.160: Rt min (method): 1.52 (a); m/z ESI+ (M+H)+: 381 [1].
Clinic trial
Background In SELECT-PsA 1, a randomised double-blind phase 3 study, upadacitinib 15 mg and 30 mg were superior to placebo and non-inferior to adalimumab in >= 20% improvement in American College of Rheumatology (ACR) criteria at 12 weeks in patients with psoriatic arthritis (PsA). Here, we report 56-week efficacy and safety in patients from SELECT-PsA 1. Methods Patients received upadacitinib 15 mg or 30 mg once daily, adalimumab 40 mg every other week for 56 weeks or placebo through week 24 switched thereafter to upadacitinib 15 mg or 30 mg until week 56. Efficacy endpoints included the proportion of patients achieving >= 20%/50%/70% improvement in ACR criteria (ACR20/50/70), >= 75%/90%/100% improvement in Psoriasis Area and Severity Index (PASI75/90/100), minimal disease activity (MDA) and change from baseline in modified total Sharp/van der Heijde Score. Treatment-emergent adverse events per 100 patient years (PY) were summarised. Results Consistent with results through week 24, ACR20/50/70, PASI75/90/100 and MDA responses were maintained with upadacitinib through week 56 and were generally numerically higher than with adalimumab; inhibition of radiographic progression was also maintained. Patients who switched from placebo to upadacitinib exhibited comparable improvements at week 56 as patients originally randomised to upadacitinib. The rates of serious adverse events were 9.1 events/100 PY with upadacitinib 15 mg and 12.3 events/100 PY with upadacitinib 30 mg. Two deaths were reported in each of the upadacitinib groups. Conclusion Efficacy across various domains of PsA were maintained with upadacitinib 15 mg and 30 mg through week 56 with no new safety signals observed [2].
Background Upadacitinib is a Janus kinase inhibitor under evaluation for the treatment of psoriatic arthritis (PsA). We evaluated upadacitinib in patients with PsA and prior inadequate response or intolerance to at least one biologic disease-modifying antirheumatic drug (DMARD).Methods In this 24-week randomised, placebo-controlled, double-blind, phase 3 trial, 642 patients were randomised (2:2:1:1) to once per day upadacitinib 15 mg or 30 mg, placebo followed by upadacitinib 15 mg or placebo followed by upadacitinib 30 mg at week 24. The primary endpoint was the proportion of patients achieving American College of Rheumatology (ACR) 20 response at week 12. Achievement of minimal disease activity (MDA) was assessed at week 24. Treatment-emergent adverse events are reported for all patients who received at least one dose of trial drug.Results At week 12, significantly more patients receiving upadacitinib 15 mg and 30 mg versus placebo achieved ACR20 (56.9% and 63.8% vs 24.1%; p<0.001 for both comparisons). At week 24, MDA was achieved by more upadacitinib 15 mg-treated (25.1%) and 30 mg-treated patients (28.9%) versus placebo (2.8%; p<0.001 for both comparisons). Generally, the rates of treatment-emergent adverse events were similar with placebo and upadacitinib 15 mg and higher with upadacitinib 30 mg at week 24. Rates of serious infections were 0.5%, 0.5% and 2.8% with placebo, upadacitinib 15 mg and upadacitinib 30 mg, respectively.Conclusion In this trial of patients with active PsA who had inadequate response or intolerance to at least one biologic DMARD, upadacitinib 15 mg and 3
You may like
Related articles And Qustion
See also
Lastest Price from Upadacitinib manufacturers

US $1.00/g2025-11-06
- CAS:
- 1310726-60-3
- Min. Order:
- 300g
- Purity:
- 99.8%
- Supply Ability:
- 20 TONS

US $100.00/G/LG2025-06-25
- CAS:
- 1310726-60-3
- Min. Order:
- 1G/LG
- Purity:
- 99%
- Supply Ability:
- 1 ton/year