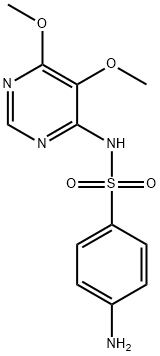
Sulfadoxine: Uses, Safety, and Resistance
The combination of sulfadoxine and pyrimethamine is used in the treatment and prophylaxis of chloroquine resistant strains of malaria and the treatment of toxoplasmosis. Sulfadoxine-pyrimethamine has been linked with rare cases of idiosyncratic liver injury which resembles the hepatotoxicity associated with sulfonamides.

Preparation of Sulfadoxine
Respectively mix DMF 175.0g, sulfa sodium formula III 106.8g (0.55mol), 87.3g (0.50mol) of 4-chloro-5,6-dimethoxypyrimidine formula II and 53.0g of sodium carbonate were added to the reaction flask. After the addition, the temperature was raised to 85-90 °C and reacted for 4 hours. After the reaction, the vacuum is turned on, and DMF is recovered by distillation under reduced pressure. After the evaporation is completed, water is added. After stirring to dissolve, adjust the pH=7-8 with dilute acetic acid, cool, filter, and the filter cake can recover the sulfonamide. Transfer the filtrate to another reaction flask, adjust the pH=5.1-5.4 with dilute acetic acid, filtered, washed, and dried to obtain 146.5 g of sulfadoxine formula I, with a yield of 94.4%; HPLC purity 99.8%, without condensate formula VI impurities.[1]
Hepatotoxicity
Sulfadoxine-pyrimethamine can cause clinically apparent, idiosyncratic liver injury with prominet features of drug-allergy or hypersensitivity, as is typical of sulfonamide hepatotoxicity. The typical onset is sudden development of fever and rash followed by jaundice within a few days or weeks of starting the medication. The pattern of injury is typically cholestatic or mixed and can be complicated and prolonged. This combination has also been linked to cases of acute liver failure with marked hepatocellular injury. However, most cases resolve rapidly, usually within 2 to 4 weeks unless cholestasis is severe. Mild ALT elevations without jaundice can accompany hypersensitivity reactions to the sulfonamides and may be accompanied by hepatic granulomas. Reexposure leads to a more rapid onset of injury and should be avoided (as should use of other sulfonamides). It is not clear whether pyrimethamine is capable of causing liver injury on its own, largely because it is used only in combination with a sulfonamide.
Sulfadoxine-pyrimethamine induced liver injury can result in acute liver failure, but most cases resolve rapidly with discontinuation of drug, and full recovery is expected within 2 to 8 weeks. Cases in which granulomatous hepatitis was identified on liver biopsy have been described, but the clinical course is not distinctive. Liver injury is usually a part of a systemic hypersensitivity reaction, and some cases can be categorized as DRESS syndome (drug rash with eosinophilia and systemic symptoms). Rechallenge should not be done, and patients should be told that they are allergic to sulfonamides (“sulfa-drugs”) and not receive other drugs in this class. Prednisone has been used with variable success, but may be particularly helpful in patients with prominent allergic features with systemic features and fever, severe rash, arthralgias, lymphadenopathy and eosinophilia or atypical lymphocytosis.[2]
Safety and toxicity of sulfadoxine/pyrimethamine
Plasmodium falciparum infection during pregnancy is strongly associated with maternal anaemia and low birth weight, contributing to substantial morbidity and mortality in sub-Saharan Africa. Randomised, controlled trials have demonstrated decreased rates of maternal anaemia and low birth weight with this approach. The WHO currently recommends IPTp-SP in malaria-endemic areas of sub-Saharan Africa. However, implementation has been suboptimal in part because of concerns of potential drug toxicities. This review evaluates the toxicity data of sulfadoxine/pyrimethamine, including severe cutaneous adverse reactions, teratogenicity and alterations in bilirubin metabolism. Weekly sulfadoxine/pyrimethamine prophylaxis is associated with rare but potentially fatal cutaneous reactions. Fortunately, sulfadoxine/pyrimethamine use in IPTp programmes in Africa, with 2-4 treatment doses over 6 months, has been well tolerated in multiple IPTp trials. However, sulfadoxine/pyrimethamine should not be administered concurrently with cotrimoxazole given their redundant mechanisms of action and synergistic worsening of adverse drug reactions. Therefore, HIV-infected pregnant women in malaria endemic areas who are already receiving cotrimoxazole prophylaxis should not also receive IPTp-SP.[3]
Although folate antagonist use in the first trimester is associated with neural tube defects, large case-control studies have demonstrated that sulfadoxine/pyrimethamine administered as IPTp (exclusively in the second and third trimesters and after organogenesis) does not result in an increased risk of teratogenesis. Folic acid supplementation is recommended for all pregnant women to reduce the rate of congenital anomalies but high doses of folic acid (5 mg/day) may interfere with the antimalarial efficacy of sulfadoxine/pyrimethamine. However, the recommended standard dose of folic acid supplementation (0.4 mg/day) does not affect antimalarial efficacy and may provide the optimal balance to prevent neural tube defects and maintain the effectiveness of IPTp-SP. No clinical association between sulfadoxine/pyrimethamine use and kernicterus has been reported despite the extensive use of sulfadoxine/pyrimethamine and related compounds to treat maternal malaria and congenital toxoplasmosis in near-term pregnant women and newborns. Although few drugs in pregnancy can be considered completely safe, sulfadoxine/pyrimethamine - when delivered as IPTp - has a favourable safety profile. Improved pharmacovigilance programmes throughout Africa are now needed to confirm its safety as access to IPTp-SP increases. Given the documented benefits of IPTp-SP in malaria endemic areas of Africa, access to this treatment for pregnant women should continue to expand.
Sulfadoxine Resistance in Plasmodium vivax
The rapid spread of chloroquine-resistant Plasmodium falciparum malaria in many countries has led to the widespread use of the sulfadoxine-pyrimethamine (S-P) combination, also known as Fansidar, for the treatment of malaria. Sulfa drugs (sulfones and sulfonamides) were first used in the 1930s for malaria treatment. Over the next decade, numerous clinical studies indicated that these drugs were usually effective against P. falciparum malaria but much less so against Plasmodium vivax malaria. With the subsequent introduction of chloroquine and its ability to rapidly control acute attacks of both P. falciparum and P. vivax malaria, little further attention was given to the slower-acting sulfa drugs. However, when chloroquine resistance started to become a problem in the early 1960s, renewed interest was shown in sulfa drugs and in exploiting the synergy of these drugs in combination with dihydrofolate reductase inhibitors. Short-acting sulfonamides soon gave way to longer-acting sulfonamides such as sulfadoxine and sulfalene. As had been shown earlier with the shorter-acting sulfonamides, the addition of pyrimethamine to sulfadoxine was appreciably more effective in curing chloroquine- and pyrimethamine-resistant P. falciparum infections than sulfadoxine alone.
Sulfadoxine is predominantly used in combination with pyrimethamine, commonly known as Fansidar, for the treatment of Plasmodium falciparum. This combination is usually less effective against Plasmodium vivax, probably due to the innate refractoriness of parasites to the sulfadoxine component. To investigate this mechanism of resistance by P. vivax to sulfadoxine, we cloned and sequenced the P. vivax dhps (pvdhps) gene. The protein sequence was determined, and three-dimensional homology models of dihydropteroate synthase (DHPS) from P. vivax as well as P. falciparum were created. The docking of it to the two DHPS models allowed us to compare contact residues in the putative sulfadoxine-binding site in both species. The predicted sulfadoxine-binding sites between the species differ by one residue, V585 in P. vivax, equivalent to A613 in P. falciparum. V585 in P. vivax is predicted by energy minimization to cause a reduction in binding of sulfadoxine to DHPS in P. vivax compared to P. falciparum. Sequencing dhps genes from a limited set of geographically different P. vivax isolates revealed that V585 was present in all of the samples, suggesting that V585 may be responsible for innate resistance of P. vivax to sulfadoxine. Additionally, amino acid mutations were observed in some P. vivax isolates in positions known to cause resistance in P. falciparum, suggesting that, as in P. falciparum, these mutations are responsible for acquired increases in resistance of P. vivax to sulfadoxine.[4]
References
[1] CHONGQING KANGLE PHARMACEUTICAL - CN112457259, 2021, A
[2] LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012-. Sulfadoxine-Pyrimethamine. [Updated 2017 Dec 5].
[3] Peters PJ, Thigpen MC, Parise ME, Newman RD. Safety and toxicity of sulfadoxine/pyrimethamine: implications for malaria prevention in pregnancy using intermittent preventive treatment. Drug Saf. 2007;30(6):481-501.
[4] Korsinczky M, Fischer K, Chen N, Baker J, Rieckmann K, Cheng Q. Sulfadoxine resistance in Plasmodium vivax is associated with a specific amino acid in dihydropteroate synthase at the putative sulfadoxine-binding site. Antimicrob Agents Chemother. 2004 Jun;48(6):2214-22.
You may like
See also
Lastest Price from Sulfadoxine manufacturers

US $88.00/Kg/Drum2025-04-21
- CAS:
- 2447-57-6
- Min. Order:
- 100KG
- Purity:
- 99%-101%; BP2015
- Supply Ability:
- 2 tons

US $100.00-75.00/kg2025-04-21
- CAS:
- 2447-57-6
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 5000