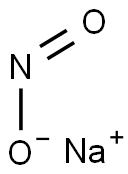Sodium nitrite: Toxicity, Application and Preparation
Indication
The Sodium nitrite[1] is a salt and an inorganic compound with the chemical formula NaNO2. It is a white to slightly yellowish crystalline powder that is very soluble in water. Sodium nitrite is commonly used as a food preservative, particularly for cured meats such as bacon, ham, and hot dogs. In addition to its use as a preservative, it is also used in the production of dyes, pharmaceuticals, and other organic compounds. However, it can be toxic if ingested in large amounts, and prolonged exposure to high levels of sodium nitrite can lead to serious health problems.

Figure 1 appearance of Sodium nitrite
Toxicity
Sodium nitrite is a class 1 carcinogen evaluated by the International Agency for Research on Cancer (lARC) (IARC, 2015) and has hematotoxicity, neurotoxicity, and immunotoxicity[2,3]. The kidney is an important target organ for the metabolic activation of exogenous chemicals in the body. Sodium nitrite can cause kidney damage, and its mechanism may be related to inhibiting the cytochrome enzyme a/a3 in renal cells, consuming intracellular GSH, GSH-Px, and SOD, inducing free radical reactions, disrupting the body's oxidative and antioxidant balance, and generating a series of chain reactions[4]. In the early stage of study, it was found that sodium nitrite has a toxic effect on hepatocytes, and the mechanism is related to cell membrane damage and oxidative stress. As a downstream target organ of the metabolic system, the kidney may be damaged by sodium nitrite toxicity, but at present, there is no report on this toxic mode and mechanism. Therefore, the study intends to take human embryonic kidney cell HEK-293 as the research object, and preliminarily explore the mechanism of sodium nitrite action on the kidney by measuring conventional renal damage indicators and oxidative damage indicators, To provide theoretical and experimental basis for exploring the molecular mechanism of sodium nitrite on the kidney.
Application
Sodium nitrite is a chemical compound that is used for a variety of purposes, including as a food preservative, a fertilizer, and in the manufacturing of dyes and other chemicals.
One of the most common uses of sodium nitrite is as a food preservative. It is often added to processed meats such as bacon, ham, and hot dogs to inhibit the growth of bacteria and prevent spoilage. Sodium nitrite also gives these products a characteristic pink color and helps to enhance their flavor.
In addition to its use in the food industry, it is also used as a corrosion inhibitor in the automotive and aerospace industries. It can help to prevent rust and other types of corrosion from forming on metal components.
Sodium nitrite has also been studied for its potential medical applications. It has been used as a treatment for cyanide poisoning and has been investigated as a possible therapy for various cardiovascular and neurological disorders. However, it can be toxic in high doses and should only be used under the guidance of a healthcare professional[5].
Measures after poisoning
Sodium nitrite poisoning can have serious effects on the body, including symptoms such as dizziness, headache, rapid heartbeat, shortness of breath, seizures, coma, and even death in severe cases. In terms of treatment, the first step is usually to remove any remaining sodium nitrite from the body. This may involve inducing vomiting or administering activated charcoal to absorb the toxin. In more severe cases, hospitalization and treatment with oxygen therapy, fluids, and medication to control symptoms may be necessary. After sodium nitrite poisoning, it is important to monitor the affected individual closely for any signs of complications or long-term effects. These may include neurological damage, kidney damage, or an increased risk of cancer. Follow-up appointments with a healthcare provider may be necessary to ensure that any ongoing effects are identified and treated appropriately.
Preparation
Sodium nitrite can be prepared by reacting sodium nitrate (NaNO3) with a reducing agent, such as sulfur dioxide (SO2), hydrogen sulfide (H2S), or iron and hydrochloric acid (Fe/ HCl). One common method for preparing sodium nitrite is the reduction of sodium nitrate using sulfur dioxide gas. The reaction takes place in an acidic solution, typically using dilute sulfuric acid (H2SO4) or hydrochloric acid (HCl) to create the necessary conditions for the reaction. The chemical equation for this reaction is: The resulting sodium nitrite can then be purified through a process such as recrystallization or fractional crystallization to remove any impurities.
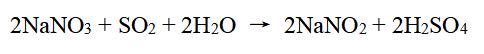
Figure 2 Chemical reaction equation of Sodium nitrite
References
[1] Manoj Kumar Shrivash,Krishna Misra. A Facile Sodium Ethoxide Mediated Knoevenagel Condensation for Production of Ethyl(E)-2-Cyano-3-Substituted Aryl/Alkyl Acrylates[J]. Proceedings of the National Academy of Sciences, India Section A: Physical Sciences,2021.
[2] Liu L C,Meng s s, Gui G, et al. Involvement of sodium nitrite in the regulation of cancer stem cell properties inhuman hepatoma cells[J].Acta Pharmaceutica Sinica, 2017,3 (2):34-35.
[3] Chu Q, Zhao Y, Shi X, et al. In vivo-like 3-D model for sodium nitrite- and acrylamide-induced hepatotoxicitytests utilizing HepG2 cells entrapped in micro-hollow fibers:[J].Scientific Reports, 2017,7(9867) :45-47.
[4] Liu M, Sun T. Effects of D-galactose, Sodium Nitrite and Alchlor on Cognitive Competence in Mice[J].ChineseJournal of Integrative Medicine on Cardio-/Cerebrovascular Disease, 2017,5(3):34-35.
[5] Sushobhan Mukhopadhyay and Sanjay Batra. Applications of Sodium Nitrite in Organic Synthesis[J]. European Journal of Organic Chemistry, 2019, 2019(38) : 6424-6451.
Related articles And Qustion
See also
Lastest Price from Sodium nitrite manufacturers

US $10.00-8.00/KG2025-04-21
- CAS:
- 7632-00-0
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt

US $9.00/KG2024-10-11
- CAS:
- 7632-00-0
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 ton