Sodium dimethyldithiocarbamate: ecotoxicity assessment and detection method
Introduction
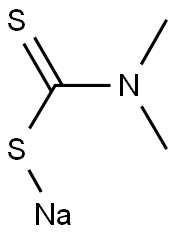
Sodium dimethyldithiocarbamate (SDDC,Figure 1), as a kind of dithiocarbamate acid (DTC), is widely used as a disinfectant, corrosion inhibitor, vulcanizing agent, chelating agent and pesticides in water treatment, rubber industry and agricultural application. Due to its excellent chelating ability, sodium dimethyldithiocarbamate has also been commercially used in harmless treatment of heavy metals such as wastewater, soil and hazardous waste treatment. Especially in China, as a chelating-stabilizing agent, sodium dimethyldithiocarbamate is extensively used in solidification/stabilization (S/S) treatment of municipal solid waste incineration (MSWI) fly ash and polluted soils.[1]

1. Ecotoxicity assessment of sodium dimethyldithiocarbamate
Sodium dimethyldithiocarbamate has very high mobility in soil and is hardly adsorbed to suspended solids or sediment. Moreover, sodium dimethyldithiocarbamate can decompose into toxic secondary products, including tetramethylthiuram, dimethylamine and thiram, as well as the major circulatory metabolites, carbon disulfide (CS2), which is a known animal and human neurotoxicant. In December 1999, there was a serious accident associated with the leakage of SDDC in which >1.5 million gallons of contaminated wastewater with sodium dimethyldithiocarbamate were discharged into local state waters, killing a total of 117 tons of fish over a 50 mile stretch from Anderson to Indianapolis, Indiana, United States.
Some studies have performed toxicological researches on sodium dimethyldithiocarbamate for different organisms, such as rabbits, zebrafish, mice and rats investigated the neurotoxicity of SDDC in mice and found that sodium dimethyldithiocarbamate resulted in motor deficits and a transient neurological deficit during the recovery period after four consecutive days of administration (1 mg/nostril) as well as caused an increase in reactive oxygen species (ROS) production within olfactory bulb and striatum. Chou et al.[2] found that SDDC selectively damaged dopaminergic (DA) neurons in organisms, and caused persistent motor deficits as well as mild injury to the nigrostriatal pathway. Several studies evidenced that SDDC exposure was related to an increased risk of Parkinson's disease (PD).
In Wang et al.'s study, the model organism Caenorhabditis elegans was used to evaluate the toxic effect of sodium dimethyldithiocarbamate and its heavy metal Cu, Pb chelates. Multiple endpoints were investigated by subacute exposure to sodium dimethyldithiocarbamate (0.01–100 mg/L) and micro-sized Cu, Pb chelates of sodium dimethyldithiocarbamate (1–100 mg/L). Our data indicated that the LC50 value of SDDC was 139.39 mg/L (95% Cl: 111.03, 174.75 mg/L). In addition, SDDC was found that concentration of 1 mg/L is a safe limit value for nematode C. elegans, and concentration above 1 mg/L caused adverse effects on the survival, growth, locomotion behaviors and reactive oxygen species (ROS) production of exposed nematodes. Furthermore, all tested SDDC-Cu and SDDC-Pb chelates had obviously lower toxic effect than untreated Cu, Pb metals. These two chelates also had a lower toxic effect than sodium dimethyldithiocarbamate agent due to its more stable structure. Moreover, SDDC-Cu had a higher toxic effect than SDDC-Pb atthe same concentration. Thus, these results suggest that sodium dimethyldithiocarbamate as a kind of chelating agent applied in harmless treatment of heavy metals, the safe addition limit should not be exceeded.[1]
2. Real-time scattered light dark-field microscopic imaging of the dynamic degradation process of sodium dimethyldithiocarbamate
Most of the investigations about the decomposition mechanism of sodium dimethyldithiocarbamate are conducted through measuring the amount of CS2 under acidic conditions, but the decomposition mechanisms of sodium dimethyldithiocarbamate under neutral and basic conditions have been neglected for a long time due to the fact that dialkyl DTCs are very stable under these conditions. It was reported that after reacting with the corresponding chlorides, the product of sodium dimethyldithiocarbamate can degrade into RSNa, Na2S, Na2CO3 and NH(CH3)2, when heated under reflux for five hours with five moles of aqueous alcoholic alkali. Nonetheless, the mechanisms of the basic decomposition of sodium dimethyldithiocarbamate were not clear until now.[3]
Conventional techniques employed in the detection of sodium dimethyldithiocarbamate, such as UV absorption spectroscopy, high-performance liquid chromatography (HPLC), and capillary electrophoresis, suffer problems of high detection limits, complicated operation and inferior efficiency. In contrast, the single nanoparticle analysis technique can be used as a new dominant platform for the detection of sodium dimethyldithiocarbamate or other pesticides.
Single nanoparticle analysis (SNA) technique with the aid of a dark-field microscopic imaging (iDFM) technique has attracted wide attention owing to its high sensitivity. Considering that the degradation of pesticides can bring about serious problems in food and the environment, and that the real-time monitoring of the dynamic degradation process of pesticides can help understand and define their degradation mechanisms, herein Gang et al. real-time monitored the decomposition dynamics of sodium dimethyldithiocarbamate under neutral and alkaline conditions by imaging single silver nanoparticles (AgNPs) under a dark-field microscope (DFM);the localized surface plasmon resonance (LSPR) scattering signals were measured at a single nanoparticle level. As a result, the chemical mechanism of the degradation of sodium dimethyldithiocarbamate under neutral and alkaline conditions was proposed, and the inhibition effects of metal ions including Zn(II) and Cu(II) were investigated in order to understand the decomposition process in different environments. It was found that Cu(II) forms the most stable complex with sodium dimethyldithiocarbamate with a stoichiometric ratio of 1:2, which greatly reduces the toxicity.
In summary, they achieved real-time monitoring of the dynamic degradation process of sodium dimethyldithiocarbamate using DFM and deduced the chemical mechanism of the degradation, expanding the SNA technique to a new field. As far as we know, it is also the first time that research on the degradation dynamics of sodium dimethyldithiocarbamate under neutral and alkaline conditions has been carried out. The reaction between sodium dimethyldithiocarbamate and AgNPs didn’t have any specific regulation, but through RGB analysis and recording the scattering spectrum before and after the reaction we developed a semi-quantitative detection method for sodium dimethyldithiocarbamate. The minimum response concentration was 10 μM, which is lower than the conventional methods. The pesticide could change the intensity as well as the color of the AgNPs, making the process of degradation visual and easy to analyze.[3]
References
[1] Wang Y, Zhang H, Wu X, et al. Ecotoxicity assessment of sodium dimethyldithiocarbamate and its micro-sized metal chelates in Caenorhabditis elegans. Sci Total Environ. 2020;720:137666. doi:10.1016/j.scitotenv.2020.137666
[2] Chou AP, Maidment N, Klintenberg R, et al. Ziram causes dopaminergic cell damage by inhibiting E1 ligase of the proteasome. J Biol Chem. 2008;283(50):34696-34703. doi:10.1074/jbc.M802210200
[3] Lei G, Gao PF, Liu H, Huang CZ. Real-time scattered light dark-field microscopic imaging of the dynamic degradation process of sodium dimethyldithiocarbamate. Nanoscale. 2015;7(48):20709-20716. doi:10.1039/c5nr05838d
See also
Lastest Price from Sodium dimethyldithiocarbamate manufacturers

US $10.00/KG2025-04-21
- CAS:
- 128-04-1
- Min. Order:
- 100KG
- Purity:
- 99%
- Supply Ability:
- 100 mt

US $99.00-35.00/kg2025-04-15
- CAS:
- 128-04-1
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 20ton