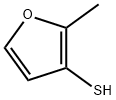
Occurrence, Aroma Contribution, Safety, and Presence in Orange Juice of 2-Methyl-3-Furanthiol
2-methyl-3-furanthiol is a metabolite found in or produced by Saccharomyces cerevisiae. 2-Methyl-3-furanthiol is a sulfur-containing compound that is the main odorant in cooked ham, wine and canned tuna. 2-Methyl-3-furanthiol is an isomer of 2FM, which exhibits a cooked meat-like aroma at an extremely low odor threshold level of 4 ng/L in a model dilute alcohol solution. Typically, the formation of which has been simultaneously investigated, because these aroma compounds are formed from the same precursor compounds (hexoses or pentoses and sulfur amino acid) by heating. 2FM has been reported to be a key aroma component of heat-treated soy sauce.

Contribution of 2-methyl-3-furanthiol to the cooked meat-like aroma
Soy sauce, a traditional fermented seasoning used throughout East Asia, is widely used on a daily basis in Japan. The aroma of soy sauce is one of the most important factors that determine its quality as well as marketability to consumers. More than 300 flavor components have been identified in Japanese soy sauce. 2-Methyl-3-furanthiol is anticipated to be the key aroma component of soy sauce, which contributes to the cooked-meat like aroma. As 2FM is generated by yeast during the fermentation of soy sauce, 2M3F is possibly generated during the same production process. For proving our hypothesis, FSS samples were compared with acid-hydrolyzed vegetable-protein-mixed soy sauce (ASS) samples in this study. Volatile thiols in the heat-treated and raw soy sauce samples were extracted and analyzed by GC-O and GC-MS. GC-O analysis with a DB-XLB column indicated cooked meat-like aroma at 20.6 min. According to the mass spectrum obtained from the same sample, the odor quality and retention time of the reference compound for 2M3F, which represents the 2-Methyl-3-furanthiol volatile thiol, was identified in the FSS sample. Kaneko et al. have reported the presence of 2M3F in raw soy sauce by GC-O, but 2M3F is not detected in the mass spectrum by GC-MS.[1]
Although no studies have been reported on the microbial or enzymatic aspects associated with the production of 2M3F, to the best of our knowledge, 2M3F is a product typically obtained from the Maillard reaction by heating. Thus, further experiments should be conducted for clarifying the mechanism for the formation of 2M3F during the fermentation of soy sauce. The cooked meat-like aroma compound, 2-methyl-3-furanthiol, was detected in fermented soy sauce (FSS) by GC-olfactometry and GC-MS. 2M3F was present in FSS at a concentration considerably greater than the perception threshold, and the 2M3F concentration increased with heating temperature. Sensory analysis indicated that with the addition of only 0.2 μg/L of 2M3F to the soy sauce sample, the cooked meat-like aroma is significantly stronger than that of sample without the addition of 2-Methyl-3-furanthiol. Hence, 2M3F contributes to the cooked meat-like aroma of FSS, which constitutes the key aroma component of FSS. In addition, 2M3F was generated from the addition of ribose and cysteine in FSS by heating at 120 °C, but it was not detected in a phosphate buffer under the same condition. Furthermore, 2M3F was not detected in acid-hydrolyzed vegetable-protein-mixed soy sauce (ASS) and heated ASS. These results indicated that fermentation by micro-organisms facilitates the generation of 2-Methyl-3-furanthiol in FSS.
Safety assessment of 2-methyl-3-furanthiol
2-Methyl-3-furanthiol was evaluated for genotoxicity, repeated dose toxicity, reproductive toxicity, local respiratory toxicity, photoirritation/photoallergenicity, skin sensitization, and environmental safety. Data show that 2-methyl-3-furanthiol is not genotoxic. The repeated dose, reproductive, and local respiratory toxicity endpoints were evaluated using the Threshold of Toxicological Concern (TTC) for a Cramer Class III material, and the exposure to 2-methyl-3-furanthiol is below the TTC (0.0015 mg/kg/day, 0.0015 mg/kg/day, and 0.47 mg/day, respectively). The skin sensitization endpoint was completed using the Dermal Sensitization Threshold (DST) for reactive materials (64 μg/cm2); exposure is below the DST. The photoirritation/photoallergenicity endpoints were evaluated based on data and ultraviolet/visible (UV/Vis) spectra; 2-methyl-3-furanthiol is not expected to be photoirritating/photoallergenic. 2-methyl-3-furanthiol was found not to be Persistent, Bioaccumulative, and Toxic (PBT) as per the International Fragrance Association (IFRA) Environmental Standards, and its risk quotients, based on its current volume of use (VoU) in Europe and North America (i.e., Predicted Environmental Concentration/Predicted No Effect Concentration [PEC/PNEC]), are <1.[2]
2-Methyl-3-furanthiol was assessed in the BlueScreen assay and found positive for cytotoxicity (positive: <80% relative cell density) and negative for genotoxicity with and without metabolic activation (RIFM, 2014). BlueScreen is a human cell-based assay for measuring the genotoxicity and cytotoxicity of chemical compounds and mixtures (Thakkar et al., 2022). Additional assays were considered to fully assess the potential mutagenic or clastogenic effects of the target material. The clastogenic activity of 2-methyl-3-furanthiol was evaluated in an in vitro micronucleus test conducted in compliance with GLP regulations and in accordance with OECD TG 487. Human peripheral blood lymphocytes were treated with 2-methyl-3-furanthiol in DMSO at concentrations of/up to 1140 μg/mL in the dose range finding (DRF) study; micronuclei analysis was conducted at concentrations up to 100 μg/mL in the presence and absence of metabolic activation. 2-Methyl-3-furanthiol did not induce binucleated cells with micronuclei when tested up to the cytotoxic concentration in either the presence or absence of an S9 activation system (
RIFM, 2021a). Under the conditions of the study, 2-methyl-3-furanthiol was considered to be non-clastogenic in the in vitro micronucleus test.
2-Methyl-3-furanthiol in Stored Orange Juice
The occurrence of methional in fresh orange juice, and possible occurrence of β-damascenone in heated orange juice, has been previously suggested. Here we report on the occurrence of 2-methyl-3-furanthiol in the headspace, collected by solid-phase micro-extraction, of fresh, pasteurized, and stored orange juice. The contents of 2-methyl-3-furanthiol and methional were quantified, and the relative level of β-damascenone was estimated, in the headspace of fresh, pasteurized, and stored orange juices using the nasal impact frequency (NIF) and surface of NIF (SNIF) GC−Olfactometry procedure. 2-Methyl-3-furanthiol concentrations were 2 ng/L in fresh and pasteurized Shamuti orange juice, and 270 ng/L in stored juice of the same variety. β-Damascenone content appeared to have increased during pasteurization and storage. Aroma-similarity experiments strongly suggest that 2-methyl-3-furanthiol and methional, at the levels found in stored orange juice (21 days at 35 °C), contribute to stored orange juice off-flavor.[3]
References
[1]Meng, Qi et al. “Contribution of 2-methyl-3-furanthiol to the cooked meat-like aroma of fermented soy sauce.” Bioscience, biotechnology, and biochemistry vol. 81,1 (2017): 168-172. doi:10.1080/09168451.2016.1238295
[2]Api, A M et al. “RIFM fragrance ingredient safety assessment, 2-methyl-3-furanthiol, CAS Registry Number 28588-74-1.” Food and chemical toxicology : an international journal published for the British Industrial Biological Research Association vol. 183 Suppl 1 (2024): 114300. doi:10.1016/j.fct.2023.114300
[3]Bezman, Y et al. “2-Methyl-3-furanthiol and methional are possible off-flavors in stored orange juice: aroma-similarity, NIF/SNIF GC-O, and GC analyses.” Journal of agricultural and food chemistry vol. 49,11 (2001): 5425-32. doi:10.1021/jf010724+
You may like
Lastest Price from 2-Methyl-3-furanthiol manufacturers

US $0.00-0.00/KG2025-06-26
- CAS:
- 28588-74-1
- Min. Order:
- 1KG
- Purity:
- 98.0%
- Supply Ability:
- 10000KGS

US $0.00/KG2025-04-15
- CAS:
- 28588-74-1
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 500000kg