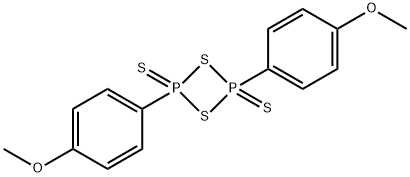
Lawesson's Reagent – a widely used agent in Organic Syntheses
Lawesson's reagent, or LR, is a chemical compound used in organic synthesis as a thiation agent. Its chemical formula is C14H14O2P2S4. Lawesson's reagent was first made popular by Sven-Olov Lawesson, who did not, however, invent it. Lawesson's reagent was first made in 1956 during a systematic study of the reactions of arenes with P4S10 [1,2].
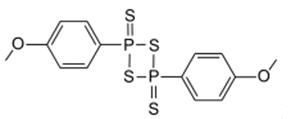
First synthesis of Lawesson's reagent (LR) appeared in 1956 along with a number of aryl thionophosphinesulfides which were the products of the reactions between P4S10 and some aromatic groups.
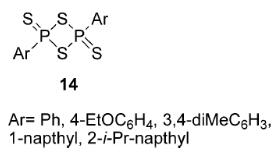
In the following decade, the chemistry of these compounds did not receive much attention. In 1967, a report appeared that LR could convert benzophenone to thiobenzophenone in acetonitrile. However, it remained unexplored for a further 10 years. In 1978, Lawesson and co-workers systematically studied the use of Lawesson’s reagent in organic syntheses, particularly for the conversion of carbonyl groups to thiocarbonyls.
Lawesson's reagent is now commercially available and widely used in organic synthesis. It can easily be synthesized with the reaction of anisole and P4S10 (150 °C, 6 h, ~70%). Also, the reaction of anisole with elemental sulfur and red phosphorus (150-155 °C, 6 h, 76%) produces LR. It was indicated that LR is not stable in solution at temperatures over 110 °C, and it decomposes or polymerizes slowly. Nishio et al. reported the reactivity order of LR toward hydroxyl and carboxyl groups. The authors indicated that hydroxyl groups are the most and esters are the least reactive functional groups among hydroxyl, amide, ketone, and esters. Amides come second and the ketones third. Their order is as follows [3]:
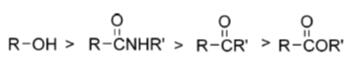
Lawesson's reagent can also be conveniently prepared in the laboratory by heating a mixture of anisole with phosphorus pentasulfide until the mixture is clear and no more hydrogen sulfide is formed [4], then recrystallized from toluene or xylene.
As Lawesson's reagent has a strong and unpleasant smell, it is best to prepare the compound within a fume-hood and to treat all glassware used with a decontamination solution before taking the glassware outside the fume-hood. One common and effective method of destroying the foul smelling residues is to use an excess of sodium hypochlorite (chlorine bleach).
Lawesson's reagent has a four membered ring of alternating sulfur and phosphorus atoms. With heating, the central phosphorus/sulfur four-membered ring can open to form two reactive dithiophosphine ylides (R-PS2). Much of the chemistry of Lawessons's reagent is in fact the chemistry of these reactive intermediates.

In general, the more electron rich a carbonyl is, the faster the carbonyl group will be converted into the corresponding thiocarbonyl by Lawesson's reagent.
Transformation of a carbonyl functional group into thiocarbonyl has been an important interest to synthetic organic chemists for many years. Lawesson’s reagent (LR) is one of the most widely used agents for such a transformation as well as for the synthesis of wide range of heterocyclic compounds having sulfur atoms. On the other hand, LR has been the most widely used reagent since the beginning of the last quarter of the 20th Century, and due to its important applications in synthetic organic chemistry, it has regularly been reviewed [3].
Lawesson's reagent effectively converts the oxo groups of ketones to thiones even in the presence of various functional groups such as aromatic and heterocyclic rings, halogen, nitro, nitrile, alkyl, alkylamine, and ester functional groups.
General Reaction of LR with Ketones:
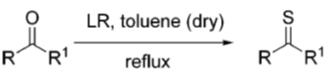
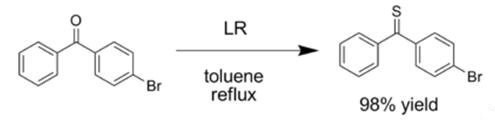
The chemistry of Lawesson's reagent and related substances has been reviewed by several groups [5]. The main use of Lawesson's reagent is the thionation of carbonyl compounds. For instance, Lawesson's reagent will convert a carbonyl into a thiocarbonyl. Additionally, Lawesson's reagent has been used to thionate enones, esters, lactones, amides, lactams, and quinones.
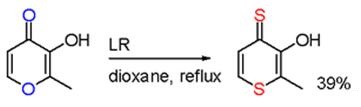
In one study, reaction of maltol with LR results in a selective oxygen replacement in two positions.
A combination of silver perchlorate and Lawesson's reagent is able to act as an oxophilic Lewis acid with the ability to catalyze the Diels-Alder reaction of dienes with α,β-unsaturated aldehydes.
Alcohols may be converted to thiols by treatment with Lawesson's reagent [1].
Lawesson's reagent reacts with sulfoxides to form a thio product, which are then desulfurized to form thioethers. Therefore, it can be used as a reducing agent for sulfoxide.
References
[1] https://en.wikipedia.org/wiki/Lawesson%27s_reagent
[2] Lecher, H. Z.; Greenwood, R. A.; Whitehouse, K. C.; Chao, T. H. (1956). "The Phosphonation of Aromatic Compounds with Phosphorus Pentasulfide". J. Am. Chem. Soc. 78 (19): 5018. doi:10.1021/ja01600a058.
[3] Turan Ozturk, Erdal Ertas, and Olcay Mert, Use of Lawesson’s Reagent in Organic Syntheses, Chem. Rev. 2007, 107, 5210-5278.
[4] Thomsen, I.; Clausen, K.; Scheibye, S.; Lawesson, S.-O. (1990). "Thiation with 2,4-Bis(4-methoxyphenyl)-1,3,2,4-Dithiadiphosphetane 2,4-disulfide: N-Methylthiopyrrolidone". Organic Syntheses.; Collective Volume, 7, p. 372
[5] Martin Jesberger; Thomas P. Davis; Leonie Barner (2003). "Applications of Lawesson's Reagent in Organic and Organometallic Syntheses". Synthesis (Review). 2003 (13): 1929–1958. doi:10.1055/s-2003-41447
You may like
Related articles And Qustion
See also
Lastest Price from Lawesson's Reagent manufacturers

US $15.00-10.00/KG2021-07-02
- CAS:
- 19172-47-5
- Min. Order:
- 1KG
- Purity:
- 99%+ HPLC
- Supply Ability:
- Monthly supply of 1 ton