Applications of Boron tribromide
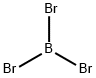
Boron tribromide, BBr3, is a colorless, fuming liquid compound containing boron and bromine. Commercial samples usually are amber to red/brown, due to weak bromine contamination. It is decomposed by water and alcohols.BBr3 is highly Lewis acidic.
It coordinates to ethereal oxygens and promotes C–O bond cleavage to an alkyl bromide and an alkoxyborane that is hydrolyzed
to an alcohol during workup.
Applications
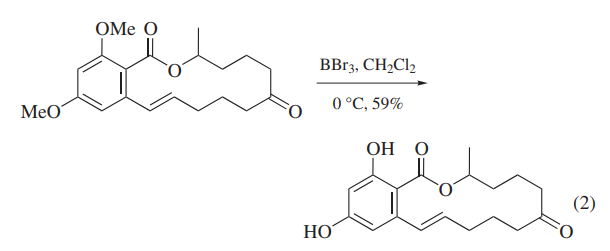
BBr3 has been widely used to cleave ethers because the reaction proceeds completely under mild conditions. In a special case, BBr3 has been used to cleave acetals that cannot be deprotected by usual acidic conditions.Because alkyl aryl ethers are cleaved at the alkyl–oxygen bond to give ArOH and alkyl bromides, BBr3 has been most generally used for the demethylation of methyl aryl ethers,for example as the final step of zearalenone synthesis (eq 2).Problems are sometimes encountered in attempts to deprotect more than one nonadjacent methoxy group on one aromatic ring, and when stable chelates are formed.6 The presence of a carbonyl substituent facilitates the selective deprotection of polymethoxyaryl compounds.
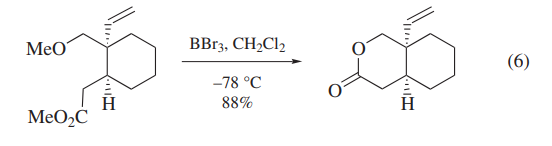
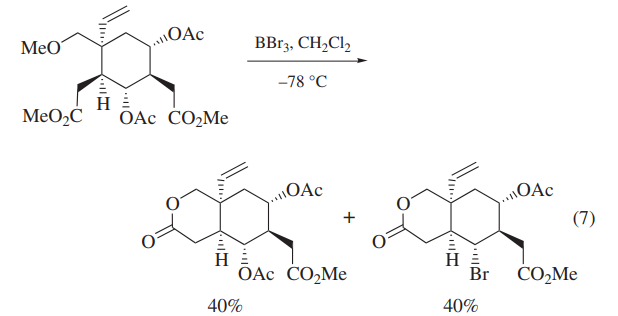
BBr3 has been also used for the deprotection of carbohydrate derivatives and polyoxygenated intermediates in the synthesis of deoxyvernolepin,vernolepin, and vernomenin.Although one of the model compounds is deprotected cleanly (eq 6),application of BBr3 to more highly functionalized intermediates leads to cleavage of undesired C–O bonds competitively (eq 7).
BBr3 can be used to effect cationic rearrangement reactions, for example, the Lewis acid-mediated ring-opening reaction and rearrangement of cyclobutyl ketones.The Lewis acid-mediated dimerization of 1,3-diarylpropargylic alcohols to give cyclobutane derivatives,the conversion of a trans-hexahydronaphthoxazine to the cis-isomer have all been achieved under the aegis of BBr3.
The BBr3-mediated conversion of 3-methoxypiperidines to 2-(bromomethyl)pyrrolidines, which proceeds via an intermediate bicyclic aziridinium ion, is an example of a rare conversion of piperidines into pyrrolidines.Renaud showed lactone ester can be obtained upon treatment of bridged bicyclic lactone with BBr3. This reaction consists of the simultaneous acidcatalyzed opening of the bicyclic ether and of the lactone.
Reference
1. McOmie, J. F. W.; Watts, M. L.; West, D. E., Tetrahedron 1968, 24,2289.
2. Meyers, A. I.; Nolen, R. L.; Collington, E. W.; Narwid, T. A.; Strickland,R. C., J. Org. Chem. 1973, 38, 1974.
3. (a) Benton, F. L.; Dillon, T. E., J. Am. Chem. Soc. 1942, 64, 1128.(b) Manson, D. L.; Musgrave, O. C., J. Chem. Soc. 1963, 1011.(c) McOmie, J. F. W.; Watts, M. L., Chem. Ind. (London) 1963, 1658.(d) Blatchly, J. M.; Gardner, D. V.; McOmie, J. F. W.; Watts, M. L., J.Chem. Soc. (C) 1968, 1545.
4. (a) Vlattas, I.; Harrison, I. T.; Tökés, L.; Fried, J. H.; Cross, A. D., J.Org. Chem. 1968, 33, 4176. (b) Taub, D.; Girotra, N. N.; Hoffsommer,R. D.; Kuo, C. H.; Slates, H. L.; Weber, S.; Wendler, N. L., Tetrahedron1968, 24, 2443.
5. (a) Stetter, H.; Wulff, C., Chem. Ber. 1960, 93, 1366. (b) Locksley, H.D.; Murray, I. G., J. Chem. Soc. (C) 1970, 392. (c) Bachelor, F. W.;Loman, A. A.; Snowdon, L. R., Synlett 1970, 48, 1554.
6. Schäfer, W.; Franck, B., Chem. Ber. 1966, 99, 160.
7. Bonner, T. G.; Bourne, E. J.; McNally, S., J. Chem. Soc. 1960, 2929.
8. Grieco, P. A.; Noguez, J. A.; Masaki, Y., J. Org. Chem. 1977, 42, 495.
9. Grieco, P. A.; Nishizawa, M.; Burke, S. D.; Marinovic, N., J. Am. Chem.Soc. 1976, 98, 1612.
10. (a) Grieco, P. A.; Hiroi, K.; Reap, J. J.; Noguez, J. A., J. Org. Chem.1975, 40, 1450. (b) Grieco, P. A.; Reap, J. J.; Noguez, J. A., Synth.Commun. 1975, 5, 155.
You may like
See also
Lastest Price from Boron tribromide manufacturers

US $100.00/bag2023-09-13
- CAS:
- 10294-33-4
- Min. Order:
- 1bag
- Purity:
- 99
- Supply Ability:
- 5000

US $120.00/KG2023-08-16
- CAS:
- 10294-33-4
- Min. Order:
- 1KG
- Purity:
- >99%
- Supply Ability:
- 50000kg/Month