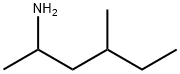
1,3-Dimethylamylamine: a stimulant similar to amphetamine
DMAA (short for 1,3-dimethylamylamine) is a stimulant similar to amphetamine and was originally developed in the 1940s as a nasal decongestant. In recent years, DMAA has been used in some dietary supplement products marketed for performance enhancement (such as pre-workouts) and weight loss. There has been some controversary around the claim that 1,3-dimethylamylamine occurs naturally in geranium plants, but according to FDA, there isn’t any reliable scientific data to support this.

The Alkylamine Stimulant 1,3-Dimethylamylamine Exhibits Substrate
The alkylamine stimulant 1,3-dimethylamylamine (DMAA) is used nonmedically as an appetite suppressant and exercise performance enhancer despite adverse cardiovascular effects that have limited its legal status. There is scant research describing the mechanism of action of 1,3-dimethylamylamine, making it difficult to gauge risks or therapeutic potential. An important molecular target of structurally related phenethylamines, such as amphetamine, for regulating mood, cognition, movement, and the development of substance use disorder is the dopamine transporter, which limits the range and magnitude of dopamine signaling via reuptake from the extracellular space. The present studies were therefore initiated to characterize the effects of DMAA on dopamine transporter function. Specifically, we tested the hypothesis that 1,3-dimethylamylamine exhibits substrate-like effects on dopamine transporter function and trafficking. In transport assays in human embryonic kidney cells, 1,3-dimethylamylamine inhibited dopamine uptake by the human dopamine transporter in a competitive manner.[1]
Docking analysis and molecular dynamics simulations supported these findings, revealing that DMAA binds to the S1 substrate binding site and induces a conformational change from outward-facing open to outward-facing closed states, similar to the known substrates. Further supporting substrate-like effects of 1,3-dimethylamylamine, the drug stimulated dopamine transporter endocytosis in a heterologous expression system via cocaine- and protein kinase A-sensitive mechanisms, mirroring findings with amphetamine. Together, these data indicate that DMAA elicits neurologic effects by binding to and regulating function of the dopamine transporter. Furthermore, pharmacologic distinctions from amphetamine reveal structural determinants for regulating transporter conformation and add mechanistic insight for the regulation of dopamine transporter endocytosis. SIGNIFICANCE STATEMENT: The alkylamine stimulant 1,3-dimethylamylamine (DMAA) is used as an appetite suppressant and athletic performance enhancer and is structurally similar to amphetamine, but there is scant research describing its mechanism of action. Characterizing the effects of 1,3-dimethylamylamine on dopamine transporter function supports evaluation of potential risks and therapeutic potential while also revealing mechanistic details of dynamic transporter-substrate interactions.
Physiological and pharmacokinetic effects of oral 1,3-dimethylamylamine
1,3-dimethylamylamine (DMAA) has been a component of dietary supplements and is also used within "party pills," often in conjunction with alcohol and other drugs. Ingestion of higher than recommended doses result in untoward effects including cerebral hemorrhage. To our knowledge, no studies have been conducted to determine both the pharmacokinetic profile and physiologic responses of 1,3-dimethylamylamine. Dietary supplements containing DMAA were once widely available, with an estimated 440,000,000 servings of such supplements sold in recent years. These doses are primarily as a component of “pre-workout” supplements marketed at those who exercise. The safety of this simple aliphatic amine has been called into question recently, partially based on case reports documented in New Zealand suggesting adverse outcomes following oral 1,3-dimethylamylamine ingestion. In these case studies, which cite cerebral hemorrhage following DMAA ingestion, individuals reported ingesting a single dose of 1,3-dimethylamylamine (for its stimulant properties), often in conjunction with caffeine and alcohol. Contrary to these case studies, several prospective investigations to date using recommended doses have not shown any untoward side effects. Despite this, DMAA has been banned in many countries, including the United States. The purpose of this study was to characterize the plasma concentration profile and associated physiological effects following a single 25 mg oral dose of 1,3-dimethylamylamine.[2]
Previously, Gee and coworkers reported a patient that purportedly ingested two “tablets” containing DMAA (later confirmed by analysis to contain 278 mg of DMAA per “capsule”: total dosage = 556 mg), along with 150 mg of caffeine and one can of beer. In a subsequent report, biochemical analysis of blood samples obtained from patients ingesting a 12.5 and 132 mg dose of DMAA indicated plasma 1,3-dimethylamylamine concentrations of 760 ng∙mL-1 (17 hours post-ingestion) and 1090 ng∙mL-1(1.66 hours post-ingestion). A third patient was noted to have a plasma DMAA concentration of 2310 ng∙mL-1 (2 hours post-ingestion); however, no information was provided regarding the ingested dosage of DMAA. As indicated in these papers, it should be noted that other chemicals may have been taken along with the 1,3-dimethylamylamine -containing products (e.g., alcohol, caffeine, phenethylamine, and cannabis).
We report for the first time the pharmacokinetic profile of oral DMAA. Based on our data, it appears that the concern over adverse health-related effects of 1,3-dimethylamylamine is specific to the dosage ingested by the individual. When ingested at recommended doses (e.g., 25 mg), our data indicate minimal to no change in heart rate, blood pressure, or body temperature, and no adverse effects were noted. We also note a consistent pattern of increase across subjects concerning peak 1,3-dimethylamylamine concentration, with mean peak values being <77 ng∙mL-1. This is approximately 15–30 times lower than plasma values reported by other investigators citing adverse outcomes following DMAA use. However, due to the case-study format of some of these adverse events, one cannot ignore possible drug interaction, errors in bioanalytical methods, differing bioavailability, or variability in exposure that might make this comparison difficult. Interpretation of our data would lead one to hypothesize that the adverse outcomes associated with 1,3-dimethylamylamine use are simply due to the blatant abuse of this ingredient. Future research on DMAA may consider pharmacokinetic characterization of each individual diastereoisomer.
Evidence for the Presence of 1,3-Dimethylamylamine in Geranium Plant Materials
1,3-Dimethylamylamine (1,3-DMAA), also known as 1,3-dimethylpentylamine and methyl hexaneamine (MHA), is an aliphatic amine with stimulant properties that is reportedly found naturally only in geranium plants (Pelargonium graveolens). The presence of 1,3-dimethylamylamine in geranium plants was first reported in a paper published in the Journal of Guizhou Institute of Technology by Ping et al. The authors detected the presence of 1,3-DMAA at a concentration of 0.66% in geranium oil isolated from fresh stems and leaves in P. graveolens plant collected from the Rongjang region of Guizhou province in China. The purpose of that study was to identify the main components of the essential oil in order to provide a basis for breed selection and to evaluate the quality of the extracted oil. However, it has since become increasingly cited as evidence for the existence of 1,3-dimethylamylamine as a natural product in certain species of geranium. In the Ping et al study, the authors used gas chromatography-mass spectrometry (GC-MS) to identify the major compounds in the oil; but some have questioned the identification of 1,3-DMAA in the oil based on apparent mislabeling of the reported chromatogram, inconsistent chromatographic elution order, and the lack of confirmation using a known standard.
1,3-Dimethylamylamine (1,3-DMAA) is an aliphatic amine with stimulant properties that are reportedly found naturally only in geranium plants (Pelargonium graveolens). The presence of 1,3-DMAA in geranium plants was first reported in a paper published in 1996, but some have questioned the identification of 1,3-dimethylamylamine in that study. Since then, a number of additional studies have been published, largely reporting the absence of 1,3-dimethylamylamine in geranium plants and commercial geranium oils. However, in two recent studies, 1,3-DMAA was detected in geranium plant tissues and a geranium oil sample using a simplified extraction approach on tissues and oil sourced from China. Whether or not 1,3-DMAA is found naturally in plants has significant implications as to how commercial products containing 1,3-dimethylamylamine are regulated by the US Food and Drug Administration. In this paper, differences in source materials, extraction procedures, and analytical approaches are reviewed in an attempt to rationalize the apparently conflicting evidence for the presence of 1,3-DMAA in geranium plant materials
References
[1]Small C, Cheng MH, Belay SS, Bulloch SL, Zimmerman B, Sorkin A, Block ER. The Alkylamine Stimulant 1,3-Dimethylamylamine Exhibits Substrate-Like Regulation of Dopamine Transporter Function and Localization. J Pharmacol Exp Ther. 2023 Aug;386(2):266-273.
[2]Schilling BK, Hammond KG, Bloomer RJ, Presley CS, Yates CR. Physiological and pharmacokinetic effects of oral 1,3-dimethylamylamine administration in men. BMC Pharmacol Toxicol. 2013 Oct 4;14:52.
[3]Whitehead PN, Schilling BK, Farney TM, Bloomer RJ. Impact of a dietary supplement containing 1,3-dimethylamylamine on blood pressure and bloodborne markers of health: a 10-week intervention study. Nutr Metab Insights. 2012 Feb 2;5:33-9.
See also
Lastest Price from 1,3-Dimethylamylamine manufacturers

US $0.00/kg2025-04-25
- CAS:
- 105-41-9
- Min. Order:
- 1kg
- Purity:
- 0.99
- Supply Ability:
- 1000kg

US $6.00/kg2025-04-21
- CAS:
- 105-41-9
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 2000KG/Month