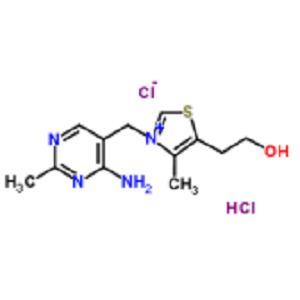
维生素B1(盐酸硫胺)
Vitamin B1 (Thiamine Hydrochloride)
67-03-8
67-03-8
询价
25KG/Carton
起订
陕西 更新日期:2024-06-28
LEADER BIOCHEMICAL GROUP XI'AN LEADER BIOCHEMICAL ENGINEERING CO.,LTD
联系人:Kevin
邮箱:sales@leader-biogroup.com
产品详情:
- 中文名称:
- 维生素B1(盐酸硫胺)
- 英文名称:
- Vitamin B1 (Thiamine Hydrochloride)
- CAS号:
- 67-03-8
- 保存条件:
- 常温避光
- 纯度规格:
- 99%
- 产品类别:
- 维生素
- color:
- white
- Assay:
- 99.9%
- Package:
- 25KG
公司简介
Since the year of 1996 Our old Leader Pharma Chemical Factory Established which is focus on manufacture & OEM Pharm biochemical products which covers the area of functional food ingredients, nutritional Ingredients, health care products, cosmetics,pharmaceutical fine chemicals,natural plant ingredients.We are keen on to provide top quality of GMP standards products as per our customers request.
For the better development and adapt to the international market ,at the year of 2014 our group integrated and headquartered in kowloon, HK, our mainly factories locates at China Mainland which is inco
| 成立日期 | (12年) |
| 注册资本 | 390万 |
| 员工人数 | 50-100人 |
| 年营业额 | ¥ 1000万-5000万 |
| 经营模式 | 工厂 |
| 主营行业 | 中间体,食品与饲料添加剂,医药原料,食品添加剂,饲料添加剂 |
维生素B1(盐酸硫胺)相关厂家报价 更多
-

- 维生素B1
- 湖北威德利化学科技有限公司 VIP
- 2026-05-21
- ¥530
-

- 维生素B1,盐酸硫胺,硝酸硫胺
- 湖北鸿福达生物科技有限公司 VIP
- 2026-05-21
- 询价
-

- 维生素B1-67-03-8
- 武汉鼎信通药业有限公司 VIP
- 2026-05-21
- 询价
-

- 维生素B1
- 湖北威德利化学试剂有限公司 VIP
- 2026-05-21
- 询价
-
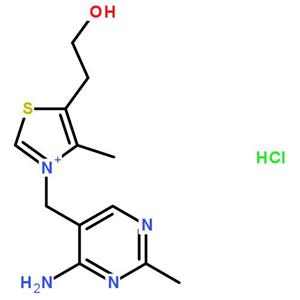
- 维生素B1(盐酸盐)
- 湖北丽都新材料科技有限公司 VIP
- 2026-05-21
- 询价
-

- 维生素B1盐酸盐 67-03-8
- 湖北鸿福达生物科技有限公司 VIP
- 2026-05-21
- 询价
-

- 维生素B1
- 南京择优新材料科技有限公司 VIP
- 2026-05-21
- 询价
-
- 维生素B1
- 上海康朗生物科技有限公司 VIP
- 2026-05-21
- ¥40
-
![维生素B1/盐酸硫胺/盐酸噻胺/硫胺盐酸盐/盐酸硫胺素/维生素B1盐酸盐/尼克酸/3-[(4-氨基-2-甲基-5-嘧啶基)甲基]-5-(2-羟乙基)-4-甲基噻唑](https://img.chemicalbook.com/SupplyImg2/2026-01-28/Large/202601281554261904209.jpg)
- 维生素B1/盐酸硫胺/盐酸噻胺/硫胺盐酸盐/盐酸硫胺素/维生素B1盐酸盐/尼克酸/3-[(4-氨基-2-甲基-5-嘧啶基)甲基]-5-(2-羟乙基)-4-甲基噻唑
- 南京都莱生物技术有限公司 VIP
- 2026-05-21
- ¥40
-

- 盐酸硫胺
- 泰安市江舟生物科技有限公司 VIP
- 2026-05-21
- 询价


